Global Cardiac Biomarkers Diagnostics Market Report: Trends, Growth and Forecast (2026-2032)
By Product (Reagents & Kits, Instruments/Analyzers, Consumables & Accessories), By Biomarker Type (Troponin (Cardiac Troponin I (cTnI), Cardiac Troponin T (cTnT), High-Sensitivity Troponin (hs-cTn)), Creatine Kinase-MB (CK-MB), Myoglobin, BNP and NT-proBNP, Other Cardiac Biomarkers), By Application (Acute Coronary Syndrome (ACS), Myocardial Infarction (MI), Congestive Heart Failure (CHF), Chest Pain/Emergency Triage, Risk Stratification & Prognosis, Other Cardiac Conditions), By Testing Location (Laboratory Testing, Point-of-Care Testing (POCT)), By End User (Hospitals, Diagnostic Laboratories, Emergency Departments, Cardiac/Specialty Clinics, Ambulatory Care Centers, Others), By Technology (Chemiluminescence Immunoassay, Fluorescence Immunoassay, ELISA, Lateral Flow/Rapid Immunoassay, Other Assay Technologies), By Sample Type (Whole Blood, Plasma, Serum), By Region (North America, South America, Europe, Middle East & Africa, Asia Pacific) ... Read more
|
Major Players
|
Global Cardiac Biomarkers Diagnostics Market Statistics and Insights, 2026
- Market Size Statistics
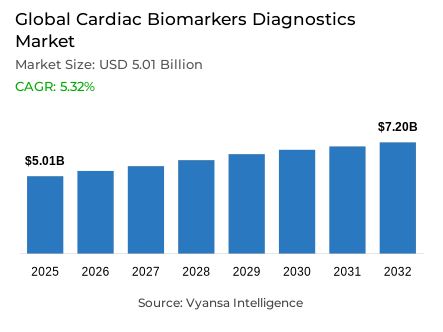
- Cardiac biomarkers diagnostics market size in Global was valued at USD 5.01 billion in 2025 and is estimated at USD 5.28 billion in 2026.
- The market size is expected to grow to USD 7.2 billion by 2032.
- Market to register a CAGR of around 5.32% during 2026-32.
- Product Shares
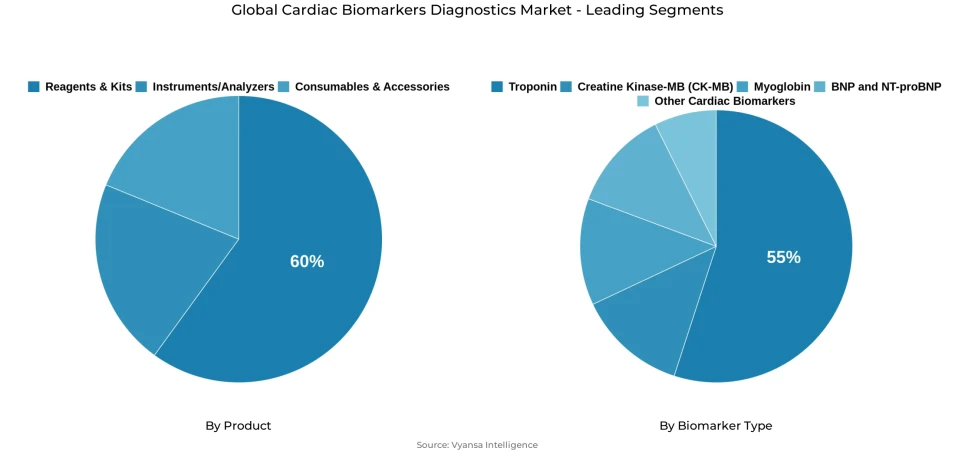
- Reagents & kits grabbed market share of 60%.
- Competition
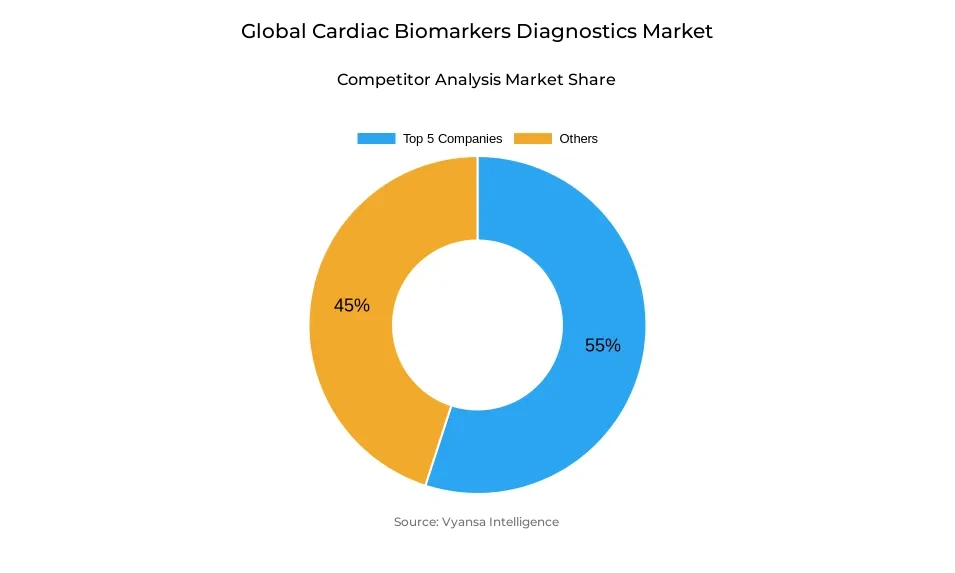
- More than 25 companies are actively engaged in producing cardiac biomarkers diagnostics.
- Top 5 companies acquired around 55% of the market share.
- Danaher Corporation, Thermo Fisher Scientific, Bio-Rad Laboratories, Roche Diagnostics, Abbott Laboratories etc., are few of the top companies.
- Biomarker Type
- Troponin grabbed 55% of the market.
- Region
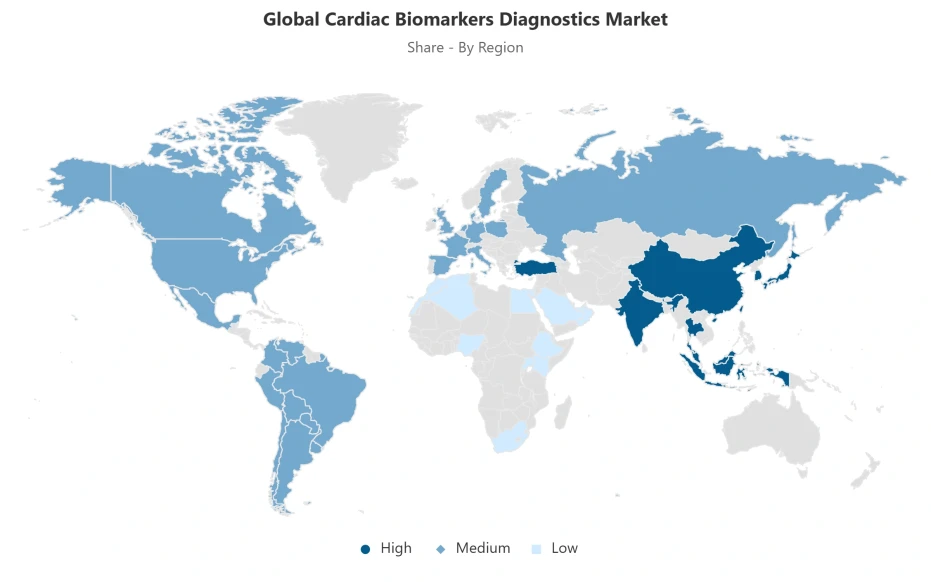
- Asia Pacific leads with a 40% share of the global market.
Global Cardiac Biomarkers Diagnostics Market Outlook
The Global cardiac biomarkers diagnostics market was valued at USD 5.01 billion in 2025, establishing a commercially resilient foundation within one of the most diagnostically critical segments of the global in-vitro diagnostics ecosystem. Projected to advance from USD 5.28 billion in 2026 to USD 7.20 billion by 2032, the sector registers a CAGR of approximately 5.32% across the forecast horizon. This steady and structurally supported expansion trajectory reflects the convergence of an escalating global cardiovascular disease burden, accelerating adoption of high-sensitivity assay platforms, and the progressive extension of cardiac biomarkers diagnostics into decentralized and point-of-care clinical environments. These forces collectively sustain consistent diagnostic volume growth across both established and emerging healthcare systems transforming cardiac biomarker testing from a specialized acute-care function into a broadly deployed, institutionally essential diagnostic infrastructure component.
The product architecture defining this market's commercial structure is characterized by pronounced concentration within the consumables category. Reagents and kits command approximately 60% of total product share a dominant position reflecting the inherently recurring nature of diagnostic consumable demand, where every test cycle across hospitals, centralized laboratories, and emergency care settings generates continuous replenishment requirements. This consumables concentration creates a commercially resilient market structure whose revenue foundation is anchored in high-frequency, operationally essential procurement activity rather than episodic capital investment decisions. The strategic implication is significant market participants with established reagent and kit portfolios benefit from a revenue generation model whose predictability and volume stability are structurally insulated from institutional budget cycle volatility.
The biomarker architecture reinforces the clinical primacy of troponin as the category's dominant testing paradigm. Troponin accounts for approximately 55% of total biomarker type market share confirming its structural centrality within acute cardiac diagnostic pathways where established clinical sensitivity, an extensive real-world validation evidence base, and deep integration into myocardial infarction rule-in and rule-out protocols make it the unambiguous reference biomarker across emergency departments, intensive care units, and acute coronary syndrome management workflows globally. The FDA's substantial equivalence decisions for high-sensitivity troponin assays including the Atellica IM High-Sensitivity Troponin I on 25 July 2024 and the i-STAT hs-TnI cartridge on 3 January 2025 validate the accelerating technological evolution of this dominant segment and confirm that regulatory momentum is actively reinforcing clinical adoption velocity.
The forward outlook through 2032 is defined by four structural market forces operating in simultaneous progression. The escalating global cardiovascular disease mortality burden documented by WHO at 19.8 million deaths in 2022 sustains institutional demand for timely cardiac diagnostic testing across healthcare systems confronting the world's leading cause of mortality. The progressive regulatory maturation of high-sensitivity assay approval pathways is expanding the clinically validated testing menu, enabling broader clinical deployment of next-generation platforms. The point-of-care testing infrastructure expansion enabled by approximately 320,000 CLIA-covered laboratory entities documented by CMS creates a vast decentralized demand absorption network whose conversion into active biomarker testing volume is being systematically enabled by regulatory approval and clinical evidence momentum. Simultaneously, certification capacity constraints created by only 19 IVDR notified bodies designated in NANDO as of December 2025 shape the competitive and compliance landscape with precision, creating differentiation advantages for participants with established regulatory management capability.
Global Cardiac Biomarkers Diagnostics Market Growth Driver
Escalating Global Cardiovascular Disease Burden Sustains Structural Diagnostic Testing Demand
The escalating and geographically pervasive cardiovascular disease mortality burden represents the primary structural driver of cardiac biomarker diagnostic demand functioning as a persistent clinical imperative that sustains institutional investment in timely rule-in and rule-out testing infrastructure across emergency departments, acute care units, and diagnostic networks confronting the world's leading cause of mortality. This disease-driven demand dynamic transcends cyclical healthcare budget fluctuations, reflecting a durable clinical necessity whose volume generation is structurally anchored in the irreducible requirement for accurate, fast cardiac event diagnosis across the global patient population. The strategic consequence for market participants is a demand floor that remains commercially robust regardless of macroeconomic cycle variation a structural characteristic that defines cardiac biomarker diagnostics as a defensively positioned market vertical within the broader in-vitro diagnostics ecosystem.
The quantitative evidence validating this demand dynamic is documented with precision by WHO. Cardiovascular diseases account for 19.8 million deaths globally in 2022 representing approximately 32% of all global deaths with 85% attributable specifically to heart attack and stroke, confirming the structural scale of the acute cardiac event burden that cardiac biomarker testing is operationally positioned to address. WHO further documents that at least 38% of the 18 million premature noncommunicable disease deaths in 2021 were caused by cardiovascular diseases, with over three-quarters of CVD deaths occurring in low- and middle-income countries establishing the geographic breadth of diagnostic demand that creates sustained institutional testing volume across diverse healthcare system contexts. These mortality metrics confirm a disease burden of sufficient scale and geographic pervasiveness to sustain structural cardiac biomarker testing demand growth through 2032.
Global Cardiac Biomarkers Diagnostics Market Challenge
Stringent Regulatory Approval Pathways Constrain New Assay Commercialization Timelines
The complexity and duration of regulatory approval processes governing cardiac biomarker diagnostic assay commercialization across major global markets represents the most consequential supply-side challenge confronting market participants creating systematic documentation, validation, and submission planning burdens that extend commercialization timelines, elevate pre-market investment requirements, and constrain the pace at which next-generation cardiac biomarker assays reach clinical deployment across healthcare settings where structural demand is expanding. For smaller and mid-tier market participants, this regulatory navigation burden creates an asymmetric competitive disadvantage relative to established diagnostics corporations with dedicated regulatory affairs infrastructure progressively concentrating market entry capability and assay approval velocity among well-resourced incumbents.
The regulatory specificity of this challenge is established with authority by the FDA and European Commission. The FDA's 510(k) pathway requires manufacturers to notify the agency at least 90 days before marketing a device imposing structured pre-submission planning obligations that compound across multi-assay development pipelines and demand sustained regulatory team investment. In Europe, only 19 IVDR notified bodies are designated in NANDO as of 2 December 2025, creating certification capacity constraints that systematically slow assay approval throughput across a market where diagnostic device volume is expanding. The first four EUDAMED modules become mandatory on 28 May 2026, further elevating the compliance architecture that cardiac diagnostic developers must navigate with precision making regulatory management capability an increasingly decisive competitive differentiator through 2032.
Unlock Market Intelligence
Explore the market potential with our data-driven report
Global Cardiac Biomarkers Diagnostics Market Trend
High-Sensitivity Troponin Assay Adoption Redefines Acute Cardiac Diagnostic Standards
The accelerating clinical adoption and regulatory validation of high-sensitivity troponin assay platforms represents the defining technological trend reshaping the cardiac biomarkers diagnostics market fundamentally elevating the analytical sensitivity threshold at which cardiac injury detection occurs and enabling earlier, more accurate acute myocardial infarction assessment that compresses clinical decision timelines, improves patient triage efficiency, and progressively displaces conventional troponin assay platforms across both centralized laboratory and point-of-care diagnostic environments. This technological transition moves the competitive differentiation axis beyond assay throughput and workflow integration into the domain of detection limit performance, rapid rule-out protocol compatibility, and point-of-care deployment capability dimensions that are redefining institutional assay selection criteria at a structural level.
The regulatory validation momentum of this trend is documented with authority by the FDA. The Atellica IM High-Sensitivity Troponin I assay receives a substantial equivalence decision on 25 July 2024 confirming regulatory acceptance of next-generation central-laboratory high-sensitivity platforms and establishing a precedent that accelerates institutional adoption confidence. The i-STAT hs-TnI cartridge with the i-STAT 1 System receives a substantial equivalence decision on 3 January 2025, extending high-sensitivity troponin capability into portable near-patient testing configurations and directly enabling emergency department and decentralized care deployment. PATHFAST hs-cTnI-II's dual indication for clinical laboratory and point-of-care settings further confirms the technological trajectory toward decentralized high-sensitivity cardiac biomarker testing creating a platform diversity that broadens clinical deployment accessibility through 2032.
Global Cardiac Biomarkers Diagnostics Market Opportunity
Point-of-Care Testing Infrastructure Expansion Creates a Scalable Decentralized Demand Base
The progressive expansion of point-of-care cardiac biomarker testing enabled by accelerating high-sensitivity assay platform miniaturization, regulatory validation of near-patient testing indications, and the vast existing decentralized healthcare facility infrastructure represents the market's most commercially scalable opportunity. This expansion provides a large and geographically distributed demand base whose conversion into active cardiac biomarker testing volume is being systematically enabled by the convergence of clinical evidence, regulatory approval, and institutional adoption momentum across emergency departments, outpatient clinics, and decentralized care settings. The commercial significance of this opportunity extends beyond volume generation point-of-care platform penetration creates new institutional relationships, reagent consumption streams, and formulary commitments that sustain long-cycle revenue generation independently of centralized laboratory procurement dynamics.
The quantitative scale of this opportunity is established by WHO and CMS with measurable precision. WHO documents that diagnostics influence approximately 70% of healthcare decisions while receiving only 3–5% of healthcare budgets confirming the structural underinvestment gap that point-of-care cardiac biomarker platform expansion is commercially positioned to address through accessible, high-impact deployment economics. CMS documents that CLIA covers approximately 320,000 laboratory entities in the United States alone establishing the vast decentralized testing infrastructure footprint within which cardiac biomarker diagnostics can expand clinical reach without requiring greenfield facility investment. Suppliers combining compact platform design, high-sensitivity assay certification, and accessible deployment economics will capture disproportionate value from this structurally significant and geographically expansive market opportunity through 2032.
Global Cardiac Biomarkers Diagnostics Market Regional Analysis
By Region
- North America
- South America
- Europe
- Middle East & Africa
- Asia Pacific
The segment with highest market share under the Region is Asia Pacific, accounting for approximately 40% of the total market. This dominant position reflects the region's structural convergence of the world's largest and most rapidly expanding patient populations, accelerating healthcare infrastructure investment across key economies, rising cardiovascular disease incidence driven by demographic aging and lifestyle epidemiological transitions, and progressive diagnostic service capacity expansion that collectively sustains Asia Pacific's position as the global market's primary demand generation geography. With two-fifths of total global market value concentrated within a single regional block, Asia Pacific defines the commercial scale, competitive intensity, and growth trajectory parameters of the global cardiac biomarkers diagnostics market making it the organizing geography around which global competitive strategy, product localization investment, and market entry prioritization decisions are constructed.
The structural dominance of Asia Pacific is anchored in the compounding interaction between the region's disproportionate share of the global cardiovascular disease burden with WHO documenting that over three-quarters of cardiovascular disease deaths occur in low- and middle-income countries where Asia Pacific economies are prominently represented and the accelerating institutional investment in diagnostic infrastructure progressively expanding testing access across previously underserved healthcare settings. As healthcare system formalization, insurance coverage expansion, and point-of-care testing platform adoption advance across the region's diverse national healthcare architectures, the commercial conditions sustaining Asia Pacific's demand leadership are intensifying rather than moderating. The region's structural position as the global market's dominant demand anchor and highest-volume growth contributor is expected to deepen through 2032.
Unlock Market Intelligence
Explore the market potential with our data-driven report
Global Cardiac Biomarkers Diagnostics Market Segmentation Analysis
By Product
- Reagents & Kits
- Instruments/Analyzers
- Consumables & Accessories
The segment with highest market share under the Product Category is Reagents & Kits, accounting for approximately 60% of the total market. This dominant position reflects the deep structural alignment between consumable product economics and the operational requirements of cardiac biomarker testing where the inherently recurring nature of reagent and kit consumption across every diagnostic test cycle generates continuous, high-frequency procurement demand that sustains a commercially stable revenue base across hospitals, reference laboratories, and emergency care settings regardless of capital equipment investment timing. With three-fifths of total market value concentrated within a single product category, reagents and kits define the entire commercial, competitive, and innovation agenda of the global cardiac biomarkers diagnostics market establishing the procurement relationships, formulary positioning dynamics, and platform dependency structures that shape institutional purchasing behavior across the entire value chain.
The structural leadership of this segment is further anchored in the demand predictability that routine clinical testing volumes create. Unlike capital equipment whose procurement follows episodic institutional budget cycles, reagent and kit consumption scales directly and consistently with diagnostic testing frequency creating a revenue generation dynamic whose commercial resilience is structurally insulated from short-cycle purchasing variability. As high-sensitivity troponin assay adoption accelerates across both centralized laboratory and point-of-care settings validated by successive FDA substantial equivalence decisions through 2024 and 2025 reagent consumption volumes are expanding in direct proportion to the broadening clinical deployment of next-generation cardiac biomarker testing platforms. Manufacturers that secure formulary inclusion within high-volume institutional testing networks will command the most defensible and durable commercial positions.
By Biomarker Type
- Troponin
- Cardiac Troponin I (cTnI)
- Cardiac Troponin T (cTnT)
- High-Sensitivity Troponin (hs-cTn)
- Creatine Kinase-MB (CK-MB)
- Myoglobin
- BNP and NT-proBNP
- Other Cardiac Biomarkers
The segment with highest market share under the Biomarker Type is Troponin, accounting for approximately 55% of the total market. This dominant position confirms troponin's structural centrality within acute cardiac diagnostic pathways where established clinical sensitivity, extensive real-world validation evidence, and deep integration into myocardial infarction, acute coronary syndrome, and heart failure diagnostic protocols make it the reference biomarker of choice across emergency departments, intensive care units, and acute care settings globally. With more than half of total market value anchored in troponin-based testing, the segment defines the clinical, commercial, and technological agenda of the global cardiac biomarkers diagnostics market establishing the assay performance benchmarks, regulatory precedent frameworks, and institutional protocol standards against which all biomarker testing innovations are evaluated.
The structural leadership of troponin is being actively reinforced by the accelerating regulatory validation of high-sensitivity assay platforms that are expanding its clinical utility beyond conventional central-laboratory applications into near-patient and point-of-care testing environments. The FDA's substantial equivalence decisions for the Atellica IM High-Sensitivity Troponin I assay on 25 July 2024 and the i-STAT hs-TnI cartridge on 3 January 2025 alongside PATHFAST hs-cTnI-II's dual clinical laboratory and point-of-care indication confirm the technological momentum broadening troponin's clinical deployment footprint. This regulatory validation cascade creates a compounding adoption dynamic each approved high-sensitivity platform increases institutional familiarity, clinical protocol integration, and formulary commitment deepening troponin's structural market dominance and reinforcing its competitive leadership.
Market Players in Global Cardiac Biomarkers Diagnostics Market
These market players maintain a significant presence in the Global cardiac biomarkers diagnostics market sector and contribute to its ongoing evolution.
- Danaher Corporation
- Thermo Fisher Scientific
- Bio-Rad Laboratories
- Roche Diagnostics
- Abbott Laboratories
- Quidel Corporation
- bioMérieux
- Siemens Healthineers
- Becton, Dickinson and Company (BD)
- Randox Laboratories
- Sysmex Corporation
- Tosoh Corporation
- PerkinElmer/Revvity
- DiaSorin
- Mindray
Market News & Updates
- Roche Diagnostics, 2025:
On September 30, 2025, Roche announced primary TSIX study results for its sixth-generation high-sensitivity Troponin T test, noting recent CE Mark approval and study validation across more than 13,000 participants, with improved sensitivity and accuracy for faster and more reliable heart attack diagnosis and more efficient emergency-department triage. This is one of the most important verified developments in the global cardiac biomarkers diagnostics market because troponin remains the category’s central clinical and commercial biomarker, and Roche’s progress raises the competitive standard for assay evidence, regulatory momentum, and acute-care workflow performance in myocardial infarction rule-in and rule-out pathways.
- QuidelOrtho, 2025:
On November 3, 2025, QuidelOrtho received FDA 510(k) clearance for its VITROS high-sensitivity Troponin I assay, with the company stating that the test expands its menu, supports myocardial infarction diagnosis, fits existing VITROS laboratory workflows, and began commercial rollout to U.S. labs later that year. The market impact is significant because it broadens access to high-sensitivity troponin testing on an established immunodiagnostic installed base, which can accelerate conversion from conventional troponin assays, intensify competition in acute cardiac testing, and improve adoption economics for hospital laboratories seeking faster cardiac decision support without changing core platform infrastructure.
Frequently Asked Questions
Related Report
- Market Segmentation
- Research Scope
- Research Methodology
- Definitions and Assumptions
- Executive Summary
- Global Cardiac Biomarkers Diagnostics Market Policies, Regulations, and Standards
- Global Cardiac Biomarkers Diagnostics Market Dynamics
- Growth Factors
- Challenges
- Trends
- Opportunities
- Global Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product
- Reagents & Kits- Market Insights and Forecast 2022-2032, USD Million
- Instruments/Analyzers- Market Insights and Forecast 2022-2032, USD Million
- Consumables & Accessories- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type
- Troponin- Market Insights and Forecast 2022-2032, USD Million
- Cardiac Troponin I (cTnI)- Market Insights and Forecast 2022-2032, USD Million
- Cardiac Troponin T (cTnT)- Market Insights and Forecast 2022-2032, USD Million
- High-Sensitivity Troponin (hs-cTn)- Market Insights and Forecast 2022-2032, USD Million
- Creatine Kinase-MB (CK-MB)- Market Insights and Forecast 2022-2032, USD Million
- Myoglobin- Market Insights and Forecast 2022-2032, USD Million
- BNP and NT-proBNP- Market Insights and Forecast 2022-2032, USD Million
- Other Cardiac Biomarkers- Market Insights and Forecast 2022-2032, USD Million
- Troponin- Market Insights and Forecast 2022-2032, USD Million
- By Application
- Acute Coronary Syndrome (ACS)- Market Insights and Forecast 2022-2032, USD Million
- Myocardial Infarction (MI)- Market Insights and Forecast 2022-2032, USD Million
- Congestive Heart Failure (CHF)- Market Insights and Forecast 2022-2032, USD Million
- Chest Pain/Emergency Triage- Market Insights and Forecast 2022-2032, USD Million
- Risk Stratification & Prognosis- Market Insights and Forecast 2022-2032, USD Million
- Other Cardiac Conditions- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location
- Laboratory Testing- Market Insights and Forecast 2022-2032, USD Million
- Point-of-Care Testing (POCT)- Market Insights and Forecast 2022-2032, USD Million
- By End User
- Hospitals- Market Insights and Forecast 2022-2032, USD Million
- Diagnostic Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Emergency Departments- Market Insights and Forecast 2022-2032, USD Million
- Cardiac/Specialty Clinics- Market Insights and Forecast 2022-2032, USD Million
- Ambulatory Care Centers- Market Insights and Forecast 2022-2032, USD Million
- Others- Market Insights and Forecast 2022-2032, USD Million
- By Technology
- Chemiluminescence Immunoassay- Market Insights and Forecast 2022-2032, USD Million
- Fluorescence Immunoassay- Market Insights and Forecast 2022-2032, USD Million
- ELISA- Market Insights and Forecast 2022-2032, USD Million
- Lateral Flow/Rapid Immunoassay- Market Insights and Forecast 2022-2032, USD Million
- Other Assay Technologies- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type
- Whole Blood- Market Insights and Forecast 2022-2032, USD Million
- Plasma- Market Insights and Forecast 2022-2032, USD Million
- Serum- Market Insights and Forecast 2022-2032, USD Million
- By Region
- North America
- South America
- Europe
- Middle East & Africa
- Asia Pacific
- By Competitors
- Competition Characteristics
- Market Share & Analysis
- By Product
- Market Size & Growth Outlook
- North America Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- By Country
- US
- Canada
- Mexico
- Rest of North America
- US Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Canada Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Mexico Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Market Size & Growth Outlook
- South America Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- By Country
- Brazil
- Argentina
- Rest of South America
- Brazil Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Argentina Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Market Size & Growth Outlook
- Europe Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- By Country
- Germany
- UK
- France
- Italy
- Spain
- Denmark
- Sweden
- Norway
- Rest of Europe
- Germany Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- UK Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- France Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Italy Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Spain Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Denmark Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Sweden Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Norway Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Market Size & Growth Outlook
- Middle East & Africa Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- By Country
- Saudi Arabia
- UAE
- Kuwait
- South Africa
- Rest of Middle East & Africa
- Saudi Arabia Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- UAE Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Kuwait Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- South Africa Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Market Size & Growth Outlook
- Asia Pacific Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- By Country
- China
- India
- Japan
- South Korea
- Australia
- Thailand
- Rest of Asia Pacific
- China Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- India Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Japan Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- South Korea Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Australia Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Thailand Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Market Size & Growth Outlook
- Competitive Outlook
- Company Profiles
- Abbott Laboratories
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Quidel Corporation
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Danaher Corporation
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Siemens Healthineers
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Roche Diagnostics
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- bioMérieux
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Thermo Fisher Scientific
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Bio-Rad Laboratories
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Becton, Dickinson and Company (BD)
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Randox Laboratories
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Sysmex Corporation
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Tosoh Corporation
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- PerkinElmer/Revvity
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- DiaSorin
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Mindray
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Abbott Laboratories
- Company Profiles
- Disclaimer
| Segment | Sub-Segment |
|---|---|
| By Product |
|
| By Biomarker Type |
|
| By Application |
|
| By Testing Location |
|
| By End User |
|
| By Technology |
|
| By Sample Type |
|
| By Region |
|
Research Methodology
This study followed a structured approach comprising four key phases to assess the size and scope of the electro-oxidation market. The process began with thorough secondary research to collect data on the target market, related markets, and broader industry context. These findings, along with preliminary assumptions and estimates, were then validated through extensive primary research involving industry experts from across the value chain. To calculate the overall market size, both top-down and bottom-up methodologies were employed. Finally, market segmentation and data triangulation techniques were applied to refine and validate segment-level estimations.
Secondary Research
The secondary research phase involved gathering data from a wide range of credible and published sources. This step helped in identifying industry trends, defining market segmentation, and understanding the market landscape and value chain.
Sources consulted during this phase included:
- Company annual reports, investor presentations, and press releases
- Industry white papers and certified publications
- Trade directories and market-recognized databases
- Articles from authoritative authors and reputable journals
- Gold and silver standard websites
Secondary research was critical in mapping out the industry's value chain and monetary flow, identifying key market segments, understanding regional variations, and tracking significant industry developments.
Other key sources:
- Financial disclosures
- Industry associations and trade bodies
- News outlets and business magazines
- Academic journals and research studies
- Paid industry databases
Primary Research
To validate secondary data and gain deeper market insights, primary research was conducted with key stakeholders across both the supply and demand sides of the market.
On the demand side, participants included decision-makers and influencers from end-user industries—such as CIOs, CTOs, and CSOs—who provided first-hand perspectives on market needs, product usage, and future expectations.
On the supply side, interviews were conducted with manufacturers, industry associations, and institutional participants to gather insights into current offerings, product pipelines, and market challenges.
Primary interviews provided critical inputs such as:
- Market size and revenue data
- Product and service breakdowns
- Market forecasts
- Regional and application-specific trends
Stakeholders consulted included:
- Leading OEM and solution providers
- Channel and distribution partners
- End users across various applications
- Independent consultants and industry specialists
Market Size Estimation and Data Triangulation
- Identifying Key Market Participants (Secondary Research)
- Goal: To identify the major players or companies in the target market. This typically involves using publicly available data sources such as industry reports, market research publications, and financial statements of companies.
- Tools: Reports from firms like Gartner, Forrester, Euromonitor, Statista, IBISWorld, and others. Public financial statements, news articles, and press releases from top market players.
- Extracting Earnings of Key Market Participants
- Goal: To estimate the earnings generated from the product or service being analyzed. This step helps in understanding the revenue potential of each market player in a specific geography.
- Methods: Earnings data can be gathered from:
- Publicly available financial reports (for listed companies).
- Interviews and primary data sources from professionals, such as Directors, VPs, SVPs, etc. This is especially useful for understanding more nuanced, internal data that isn't publicly disclosed.
- Annual reports and investor presentations of key players.
- Data Collation and Development of a Relevant Data Model
- Goal: To collate inputs from both primary and secondary sources into a structured, data-driven model for market estimation. This model will incorporate key market KPIs and any independent variables relevant to the market.
- Key KPIs: These could include:
- Market size, growth rate, and demand drivers.
- Industry-specific metrics like market share, average revenue per customer (ARPC), or average deal size.
- External variables, such as economic growth rates, inflation rates, or commodity prices, that could affect the market.
- Data Modeling: Based on this data, the market forecasts are developed for the next 5 years. A combination of trend analysis, scenario modeling, and statistical regression might be used to generate projections.
- Scenario Analysis
- Goal: To test different assumptions and validate how sensitive the market is to changes in key variables (e.g., market demand, regulatory changes, technological disruptions).
- Types of Scenarios:
- Base Case: Based on current assumptions and historical data.
- Best-Case Scenario: Assuming favorable market conditions, regulatory environments, and technological advancements.
- Worst-Case Scenario: Accounting for adverse factors, such as economic downturns, stricter regulations, or unexpected disruptions.