Southeast Asia Laboratory Information Management System Market Report: Trends, Growth and Forecast (2026-2032)
By Product Type (Clinical Operations LIMS (Hospital Laboratory LIMS, Physician Office Laboratory LIMS, Routine Diagnostics Laboratory LIMS), High-Throughput Testing LIMS (Independent Laboratory LIMS, Commercial Laboratory LIMS, Reference Laboratory LIMS, Multi-site Laboratory Network LIMS), Public Health & Reporting LIMS (Public Health Laboratory LIMS, Surveillance-linked Laboratory LIMS, Reference Network Laboratory LIMS), Research & Repository LIMS (Academic Research Laboratory LIMS, Translational Research Laboratory LIMS, Biobank & Repository LIMS), Regulated QA/QC LIMS (Pharmaceutical Laboratory LIMS, Biotechnology Laboratory LIMS, Food Testing Laboratory LIMS, Environmental Testing Laboratory LIMS, Forensic Laboratory LIMS)), By Workflow Type (Accessioning-to-Result Management (Sample Registration, Sample Tracking, Test Assignment, Result Entry & Release), Instrument-Integrated Workflow Management (Analyzer Connectivity, Automated Data Capture, Bidirectional Instrument Communication), Quality-Controlled Laboratory Workflow (Quality Control Management, Quality Assurance Workflow, Exception & Deviation Handling), Traceability & Repository Workflow (Chain-of-Custody Tracking, Storage & Retrieval Management, Biobank Sample Lifecycle Tracking), Interoperability & Reporting Workflow (External System Connectivity, Standards-based Data Exchange, Regulatory & Public Health Reporting)), By Deployment Model (Controlled Environment Deployment, Scalable Digital Deployment, Mixed Infrastructure Deployment), By End-User Cluster (Care Delivery Laboratories (Hospital Laboratories, Health-System Laboratories, Physician Office Laboratories, Community & Decentralized Testing Laboratories), Commercial Testing Laboratories (Independent Laboratories, Commercial Laboratories, Reference Laboratories), Government & Public Health Laboratories (Public Health Laboratories, Central Reference Laboratories, Disease Surveillance Laboratories), Research & Translational Laboratories (Academic Laboratories, Research Institutes, Biobanks, Clinical Research Support Laboratories), Regulated Scientific & Industrial Laboratories (Pharmaceutical Laboratories, Biotechnology Laboratories, Food Safety Laboratories, Environmental Laboratories, Forensic Laboratories)), By Purchase Model (Perpetual License, Subscription License, Usage-Based Model, Hybrid Commercial Model), By Country (Indonesia, Thailand, Vietnam, Malaysia, Philippines, Singapore, Rest of Southeast Asia) ... Read more
|
Major Players
|
Southeast Asia Laboratory Information Management System Market Statistics and Insights, 2026
- Market Size Statistics
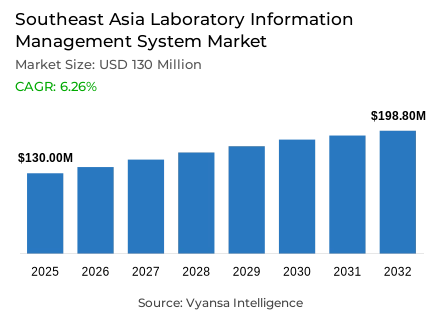
- Laboratory information management system market size in Southeast Asia was valued at USD 130 million in 2025 and is estimated at USD 138.1 million in 2026.
- The market size is expected to grow to USD 198.8 million by 2032.
- Market to register a CAGR of around 6.26% during 2026-32.
- Product Type Shares
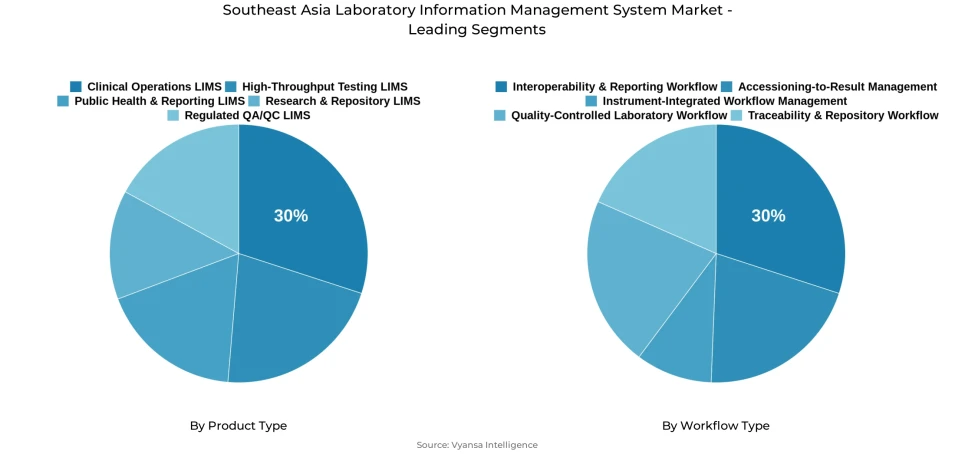
- Clinical operations lims grabbed market share of 30%.
- Competition
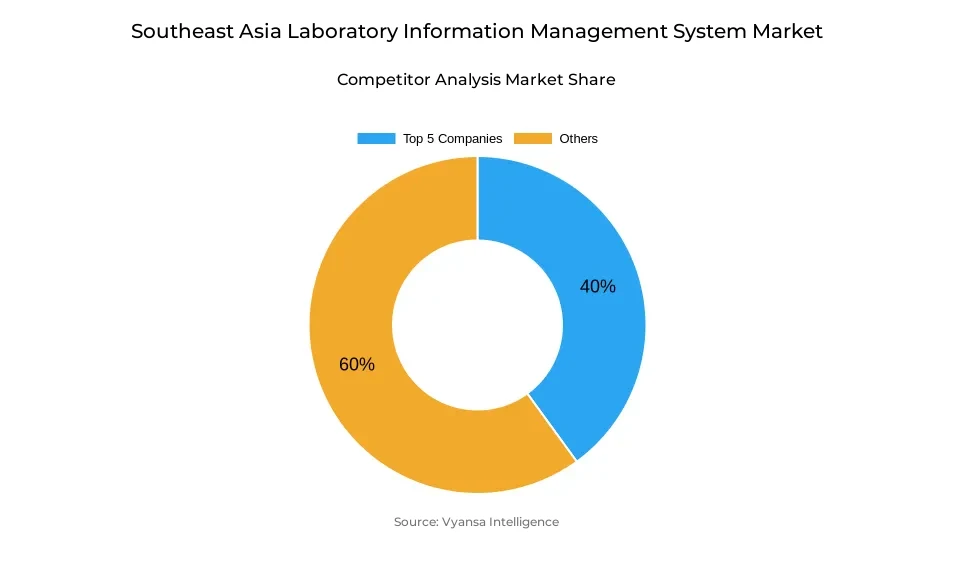
- More than 10 companies are actively engaged in producing laboratory information management system in Southeast Asia.
- Top 5 companies acquired around 40% of the market share.
- STARLIMS, Sapio Sciences, CloudLIMS, LabWare, Thermo Fisher Scientific etc., are few of the top companies.
- Workflow Type
- Interoperability & Reporting Workflow grabbed 30% of the market.
- Country
- Indonesia leads with a 30% share of the Southeast Asia market.
Southeast Asia Laboratory Information Management System Market Outlook
Southeast Asia laboratory information management system market was valued at USD 130 million in 2025 and is projected to grow from USD 138.1 million in 2026 to USD 198.8 million by 2032, registering a CAGR of 6.26% across the forecast period. Underpinning this trajectory are LIMS market growth trends driven by rising demand for better digital control of laboratory data, stronger sample traceability, and smoother reporting across clinical, diagnostic, and research settings throughout the region.
At the product level, Clinical Operations LIMS holds 30% of the overall market share, reflecting sustained laboratory demand for solutions that support routine workflows, record management, and operational visibility. The deployment of sample tracking and management systems across diagnostic and clinical environments is being treated as a foundational requirement as laboratories across Southeast Asia manage growing testing complexity and expect more organized daily execution across their core operations.
Workflow demand is shaped equally by the Interoperability and Reporting Workflow segment, which accounts for 30% of the overall market. The growing need to connect instruments, systems, and reporting channels with fewer manual steps and better data consistency is reinforcing the commercial relevance of web-based LIMS platforms that support smoother coordination, reduced duplication, and more reliable reporting structures across the region's diverse laboratory network.
Country-level demand is anchored by Indonesia, which leads the Southeast Asia market with a 30% share, reflecting strong digital health momentum and broader health system integration efforts underway across Indonesia. With product demand, workflow priorities, and country-level concentration all moving in a commercially favorable direction, the laboratory management software market across Southeast Asia is expected to strengthen its role in laboratory efficiency, reporting discipline, and connected data management through 2032.
Southeast Asia Laboratory Information Management System Market Growth Driver
National Health Data Integration Accelerates Adoption
Regional policy momentum is creating a strong and structured demand base for laboratory informatics systems across Southeast Asia. As per data published by the World Health Organization South-East Asia Regional Office, at least 80% of the countries in the region have drafted or completed national digital health strategies, establishing a stronger foundation for interoperability, health information system upgrades, and structured data exchange across clinical and laboratory settings. This policy environment is accelerating digital transformation in laboratories as institutions are expected to manage sample records, testing results, and reporting flows within wider national digital health architectures.
Country-level execution is translating this regional momentum into tangible adoption demand. According to statistics released by Indonesia's Ministry of Health, 34,463 healthcare facilities had been integrated with the SATUSEHAT platform as of 27 October 2025, with the system designed to unify patient data across hospitals, clinics, laboratories, and pharmacies into a single national health information environment. As more facilities connect to these national platforms, laboratory workflow automation software is becoming an operational necessity for laboratories that must deliver traceable records, standardized data, and faster result exchange at scale.
Southeast Asia Laboratory Information Management System Market Challenge
Fragmented Infrastructure Slows Consistent Deployment
Uneven digital readiness across Southeast Asia's healthcare network is creating a persistent structural barrier to broad and consistent laboratory informatics deployment. Based on data from the World Health Organization South-East Asia Regional Office, countries in the region continue to face fragmented and siloed health information systems, limited interoperability, technology lock-in, and inadequate scalable digital infrastructure. These conditions directly complicate data integrity and cybersecurity in labs management, as laboratories must connect new software with legacy workflows that were not designed for shared or standardized data exchange environments.
The challenge is reinforced at the country implementation level across the region. As indicated by authoritative sources at Indonesia's Ministry of Health, significant challenges remain across Indonesia's regions in areas including infrastructure and human resource readiness, even as 34,463 healthcare facilities have been integrated into SATUSEHAT. These operational realities mean that deployment timelines for regulatory compliance software for labs are frequently extended by connectivity gaps, inconsistent standards, and the complexity of managing change across dispersed, resource-constrained laboratory networks in Southeast Asia.
Unlock Market Intelligence
Explore the market potential with our data-driven report
Southeast Asia Laboratory Information Management System Market Trend
Standards-Based Interoperability Defines the Next Phase
Structured, standards-based data exchange is emerging as the defining operational expectation across Southeast Asia's evolving laboratory and healthcare information environment. Evidence drawn from public data released by Enterprise Singapore confirms that two new standards were launched in October 2025, specifically SS719:2025 for terminology standards supporting interoperability of healthcare system records and SS720:2025 for remote clinical monitoring. This signals a regional shift beyond simple digitization toward more structured data environments where laboratory information is easier to exchange and interpret, reinforcing demand for cloud-based LIMS solutions that operate within these emerging regional standards frameworks.
The trend is becoming more operationally embedded in connected care delivery. In line with findings from Singapore's Ministry of Health, all nine private hospitals in Singapore committed in November 2024 to contribute patient health information to the National Electronic Health Record, with the shared record encompassing diagnoses, medications, allergies, radiological reports, and laboratory results. This integration strengthens the institutional case for ELN integration and connected laboratory platforms that can deliver cleaner data standards, smoother reporting, and stronger coordination with regional health information ecosystems across Southeast Asia.
Southeast Asia Laboratory Information Management System Market Opportunity
Genomics and Molecular Programs Expand the Market Runway
Southeast Asia's expanding genomics and molecular testing ecosystem is generating a commercially significant opportunity for advanced laboratory informatics solutions across the region. As per official figures from Singapore's Ministry of Health, the Familial Hypercholesterolaemia genetic testing programme was formally launched on 30 June 2025, with three Genomic Assessment Centres being established and the condition estimated to affect approximately 20,000 people in Singapore alone. As genetic testing becomes more structured and program-linked, AI and machine learning in LIMS platforms are gaining relevance for managing referral data, sample movement, results processing, and connected reporting across multi-site genomic workflows.
The opportunity is widening across the broader regional network beyond Singapore. Insights sourced from official databases at the World Health Organization South-East Asia Regional Office indicate that nine of the region's ten Member States are endemic for dengue, and a five-day regional workshop on dengue genome sequencing and bioinformatics was conducted in March 2026 to strengthen genomic surveillance capacity. As molecular workflows expand across public health and research settings in Southeast Asia, demand for scalable laboratory management systems capable of supporting standardized reporting, multi-site coordination, and high-volume data management is expected to grow meaningfully through the forecast period.
Southeast Asia Laboratory Information Management System Market Country Analysis
By Country
- Indonesia
- Thailand
- Vietnam
- Malaysia
- Philippines
- Singapore
- Rest of Southeast Asia
Indonesia holds the strongest country-level position within the Southeast Asia laboratory information management system market at a 30% share, establishing it as the most commercially significant contributor to regional growth. This leadership is directly supported by Indonesia's national health data integration agenda, as evidenced by the SATUSEHAT platform's design to unify patient data across hospitals, clinics, laboratories, and pharmacies into a single connected health information environment. Indonesia's position reflects exactly the institutional conditions that drive demand for emerging markets for LIMS adoption solutions, combining policy ambition with large-scale facility integration at national level.
The scale of Indonesia's digital health execution reinforces its regional leadership further. As per data published by Indonesia's Ministry of Health, 34,463 healthcare facilities had been integrated with SATUSEHAT by 27 October 2025, with the broader SATUSEHAT ecosystem encompassing medical records, prescriptions, logistics, claims, and workforce modules across a large and geographically distributed network. As this infrastructure matures, demand for LIMS in biotechnology and clinical laboratory settings across Indonesia is expected to deepen, reinforcing the country's role as the primary growth engine within the Southeast Asia laboratory information management system market through the forecast period.
Unlock Market Intelligence
Explore the market potential with our data-driven report
Southeast Asia Laboratory Information Management System Market Segmentation Analysis
By Product Type
- Clinical Operations LIMS
- Hospital Laboratory LIMS
- Physician Office Laboratory LIMS
- Routine Diagnostics Laboratory LIMS
- High-Throughput Testing LIMS
- Independent Laboratory LIMS
- Commercial Laboratory LIMS
- Reference Laboratory LIMS
- Multi-site Laboratory Network LIMS
- Public Health & Reporting LIMS
- Public Health Laboratory LIMS
- Surveillance-linked Laboratory LIMS
- Reference Network Laboratory LIMS
- Research & Repository LIMS
- Academic Research Laboratory LIMS
- Translational Research Laboratory LIMS
- Biobank & Repository LIMS
- Regulated QA/QC LIMS
- Pharmaceutical Laboratory LIMS
- Biotechnology Laboratory LIMS
- Food Testing Laboratory LIMS
- Environmental Testing Laboratory LIMS
- Forensic Laboratory LIMS
Clinical Operations LIMS commands the highest share within the product type category at 30%, reflecting consistent institutional demand for platforms that manage routine operations, day-to-day records, and internal process visibility across clinical and diagnostic settings in Southeast Asia. Laboratories are actively investing in systems that improve operational discipline and reduce workflow inconsistencies, with LIMS for clinical diagnostics laboratories representing one of the most commercially active deployment areas across the region's healthcare network. Its leading position confirms that routine execution reliability remains the most important value driver in product selection decisions.
This segment's strength reflects a market-wide preference for solutions that improve structured handling of workflows, records, and laboratory activities rather than addressing only narrow digital functions. Lab inventory management systems capabilities embedded within Clinical Operations platforms are adding commercial depth as laboratories seek better control over reagent tracking, equipment usage, and consumable management alongside core workflow functions. As laboratories across Southeast Asia continue to professionalize and standardize their operations, this segment is expected to maintain its leading position through the forecast period.
By Workflow Type
- Accessioning-to-Result Management
- Sample Registration
- Sample Tracking
- Test Assignment
- Result Entry & Release
- Instrument-Integrated Workflow Management
- Analyzer Connectivity
- Automated Data Capture
- Bidirectional Instrument Communication
- Quality-Controlled Laboratory Workflow
- Quality Control Management
- Quality Assurance Workflow
- Exception & Deviation Handling
- Traceability & Repository Workflow
- Chain-of-Custody Tracking
- Storage & Retrieval Management
- Biobank Sample Lifecycle Tracking
- Interoperability & Reporting Workflow
- External System Connectivity
- Standards-based Data Exchange
- Regulatory & Public Health Reporting
The Interoperability and Reporting Workflow segment holds the highest share within the workflow type category at 30%, driven by growing laboratory demand for systems that help data move more smoothly across instruments, teams, and reporting endpoints with fewer manual steps and better consistency. Laboratories across Southeast Asia are prioritizing workflows that reduce duplication, improve communication between systems, and support more organized result outputs, with lab data analytics and reporting tools being deployed as a core component of reporting-first adoption strategies. This segment's leadership confirms that connected reporting quality is a primary operational priority across the region's laboratory landscape.
Its commercial dominance reflects a deeper institutional shift in how laboratories approach digital upgrade planning across Southeast Asia. Reporting continuity and cross-system coordination are no longer treated as secondary benefits but as foundational requirements for platform selection. The growing adoption of smart laboratories and automation principles across the region is reinforcing this expectation, as laboratories increasingly require workflow platforms that treat interoperability as a built-in capability rather than a configuration add-on. As these priorities intensify, this segment is expected to sustain its position as the most strategically significant workflow category within the Southeast Asia laboratory information management system market.
Various Market Players in Southeast Asia Laboratory Information Management System Market
The companies mentioned below are highly active in the Southeast Asia laboratory information management system market, occupying a considerable portion of the market and shaping industry progress.
- STARLIMS
- Sapio Sciences
- CloudLIMS
- LabWare
- Thermo Fisher Scientific
- LabVantage Solutions
- Agilent Technologies
- Autoscribe Informatics
- QBench
- SciSure
- Dassault Systèmes BIOVIA
- LabLynx
Market News & Updates
- LabVantage Solutions, 2026:
LabVantage launched LabVantage CORTEX, extending its LIMS platform with agentic AI, analytics, and cloud-native automation. The release includes an orchestration layer for laboratory workflows and was introduced with features covering automation, data insight, IoT connectivity, and digital twin integration across laboratory environments.
- CloudLIMS, 2026:
CloudLIMS released version 4.90 of its laboratory information management system. The update introduced faster sample searches, analyte-based result flagging, client shipment tracking, and training expiry alerts, adding new workflow, traceability, and compliance-related functions to the company’s cloud-based LIMS platform.
Frequently Asked Questions
Related Report
- Market Segmentation
- Research Scope
- Research Methodology
- Definitions and Assumptions
- Executive Summary
- South East Asia Laboratory Information Management System Market Policies, Regulations, and Standards
- South East Asia Laboratory Information Management System Market Dynamics
- Growth Factors
- Challenges
- Trends
- Opportunities
- South East Asia Laboratory Information Management System Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product Type
- Clinical Operations LIMS- Market Insights and Forecast 2022-2032, USD Million
- Hospital Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Physician Office Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Routine Diagnostics Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- High-Throughput Testing LIMS- Market Insights and Forecast 2022-2032, USD Million
- Independent Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Commercial Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Reference Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Multi-site Laboratory Network LIMS- Market Insights and Forecast 2022-2032, USD Million
- Public Health & Reporting LIMS- Market Insights and Forecast 2022-2032, USD Million
- Public Health Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Surveillance-linked Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Reference Network Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Research & Repository LIMS- Market Insights and Forecast 2022-2032, USD Million
- Academic Research Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Translational Research Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Biobank & Repository LIMS- Market Insights and Forecast 2022-2032, USD Million
- Regulated QA/QC LIMS- Market Insights and Forecast 2022-2032, USD Million
- Pharmaceutical Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Biotechnology Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Food Testing Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Environmental Testing Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Forensic Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Clinical Operations LIMS- Market Insights and Forecast 2022-2032, USD Million
- By Workflow Type
- Accessioning-to-Result Management- Market Insights and Forecast 2022-2032, USD Million
- Sample Registration- Market Insights and Forecast 2022-2032, USD Million
- Sample Tracking- Market Insights and Forecast 2022-2032, USD Million
- Test Assignment- Market Insights and Forecast 2022-2032, USD Million
- Result Entry & Release- Market Insights and Forecast 2022-2032, USD Million
- Instrument-Integrated Workflow Management- Market Insights and Forecast 2022-2032, USD Million
- Analyzer Connectivity- Market Insights and Forecast 2022-2032, USD Million
- Automated Data Capture- Market Insights and Forecast 2022-2032, USD Million
- Bidirectional Instrument Communication- Market Insights and Forecast 2022-2032, USD Million
- Quality-Controlled Laboratory Workflow- Market Insights and Forecast 2022-2032, USD Million
- Quality Control Management- Market Insights and Forecast 2022-2032, USD Million
- Quality Assurance Workflow- Market Insights and Forecast 2022-2032, USD Million
- Exception & Deviation Handling- Market Insights and Forecast 2022-2032, USD Million
- Traceability & Repository Workflow- Market Insights and Forecast 2022-2032, USD Million
- Chain-of-Custody Tracking- Market Insights and Forecast 2022-2032, USD Million
- Storage & Retrieval Management- Market Insights and Forecast 2022-2032, USD Million
- Biobank Sample Lifecycle Tracking- Market Insights and Forecast 2022-2032, USD Million
- Interoperability & Reporting Workflow- Market Insights and Forecast 2022-2032, USD Million
- External System Connectivity- Market Insights and Forecast 2022-2032, USD Million
- Standards-based Data Exchange- Market Insights and Forecast 2022-2032, USD Million
- Regulatory & Public Health Reporting- Market Insights and Forecast 2022-2032, USD Million
- Accessioning-to-Result Management- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model
- Controlled Environment Deployment- Market Insights and Forecast 2022-2032, USD Million
- Scalable Digital Deployment- Market Insights and Forecast 2022-2032, USD Million
- Mixed Infrastructure Deployment- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster
- Care Delivery Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Hospital Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Health-System Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Physician Office Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Community & Decentralized Testing Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Commercial Testing Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Independent Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Commercial Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Reference Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Government & Public Health Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Public Health Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Central Reference Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Disease Surveillance Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Research & Translational Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Academic Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Research Institutes- Market Insights and Forecast 2022-2032, USD Million
- Biobanks- Market Insights and Forecast 2022-2032, USD Million
- Clinical Research Support Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Regulated Scientific & Industrial Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Pharmaceutical Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Biotechnology Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Food Safety Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Environmental Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Forensic Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Care Delivery Laboratories- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model
- Perpetual License- Market Insights and Forecast 2022-2032, USD Million
- Subscription License- Market Insights and Forecast 2022-2032, USD Million
- Usage-Based Model- Market Insights and Forecast 2022-2032, USD Million
- Hybrid Commercial Model- Market Insights and Forecast 2022-2032, USD Million
- By Country
- Indonesia
- Thailand
- Vietnam
- Malaysia
- Philippines
- Singapore
- Rest of Southeast Asia
- By Competitors
- Competition Characteristics
- Market Share & Analysis
- By Product Type
- Market Size & Growth Outlook
- Indonesia Laboratory Information Management System Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product Type- Market Insights and Forecast 2022-2032, USD Million
- By Workflow Type- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Thailand Laboratory Information Management System Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product Type- Market Insights and Forecast 2022-2032, USD Million
- By Workflow Type- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Vietnam Laboratory Information Management System Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product Type- Market Insights and Forecast 2022-2032, USD Million
- By Workflow Type- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Malaysia Laboratory Information Management System Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product Type- Market Insights and Forecast 2022-2032, USD Million
- By Workflow Type- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Philippines Laboratory Information Management System Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product Type- Market Insights and Forecast 2022-2032, USD Million
- By Workflow Type- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Singapore Laboratory Information Management System Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product Type- Market Insights and Forecast 2022-2032, USD Million
- By Workflow Type- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Competitive Outlook
- Company Profiles
- LabWare
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Thermo Fisher Scientific
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- LabVantage Solutions
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Agilent Technologies
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Autoscribe Informatics
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- STARLIMS
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Sapio Sciences
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- CloudLIMS
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- QBench
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- SciSure
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Dassault Systèmes BIOVIA
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- LabLynx
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- LabWare
- Company Profiles
- Disclaimer
| Segment | Sub-Segment |
|---|---|
| By Product Type |
|
| By Workflow Type |
|
| By Deployment Model |
|
| By End-User Cluster |
|
| By Purchase Model |
|
| By Country |
|
Research Methodology
This study followed a structured approach comprising four key phases to assess the size and scope of the electro-oxidation market. The process began with thorough secondary research to collect data on the target market, related markets, and broader industry context. These findings, along with preliminary assumptions and estimates, were then validated through extensive primary research involving industry experts from across the value chain. To calculate the overall market size, both top-down and bottom-up methodologies were employed. Finally, market segmentation and data triangulation techniques were applied to refine and validate segment-level estimations.
Secondary Research
The secondary research phase involved gathering data from a wide range of credible and published sources. This step helped in identifying industry trends, defining market segmentation, and understanding the market landscape and value chain.
Sources consulted during this phase included:
- Company annual reports, investor presentations, and press releases
- Industry white papers and certified publications
- Trade directories and market-recognized databases
- Articles from authoritative authors and reputable journals
- Gold and silver standard websites
Secondary research was critical in mapping out the industry's value chain and monetary flow, identifying key market segments, understanding regional variations, and tracking significant industry developments.
Other key sources:
- Financial disclosures
- Industry associations and trade bodies
- News outlets and business magazines
- Academic journals and research studies
- Paid industry databases
Primary Research
To validate secondary data and gain deeper market insights, primary research was conducted with key stakeholders across both the supply and demand sides of the market.
On the demand side, participants included decision-makers and influencers from end-user industries—such as CIOs, CTOs, and CSOs—who provided first-hand perspectives on market needs, product usage, and future expectations.
On the supply side, interviews were conducted with manufacturers, industry associations, and institutional participants to gather insights into current offerings, product pipelines, and market challenges.
Primary interviews provided critical inputs such as:
- Market size and revenue data
- Product and service breakdowns
- Market forecasts
- Regional and application-specific trends
Stakeholders consulted included:
- Leading OEM and solution providers
- Channel and distribution partners
- End users across various applications
- Independent consultants and industry specialists
Market Size Estimation and Data Triangulation
- Identifying Key Market Participants (Secondary Research)
- Goal: To identify the major players or companies in the target market. This typically involves using publicly available data sources such as industry reports, market research publications, and financial statements of companies.
- Tools: Reports from firms like Gartner, Forrester, Euromonitor, Statista, IBISWorld, and others. Public financial statements, news articles, and press releases from top market players.
- Extracting Earnings of Key Market Participants
- Goal: To estimate the earnings generated from the product or service being analyzed. This step helps in understanding the revenue potential of each market player in a specific geography.
- Methods: Earnings data can be gathered from:
- Publicly available financial reports (for listed companies).
- Interviews and primary data sources from professionals, such as Directors, VPs, SVPs, etc. This is especially useful for understanding more nuanced, internal data that isn't publicly disclosed.
- Annual reports and investor presentations of key players.
- Data Collation and Development of a Relevant Data Model
- Goal: To collate inputs from both primary and secondary sources into a structured, data-driven model for market estimation. This model will incorporate key market KPIs and any independent variables relevant to the market.
- Key KPIs: These could include:
- Market size, growth rate, and demand drivers.
- Industry-specific metrics like market share, average revenue per customer (ARPC), or average deal size.
- External variables, such as economic growth rates, inflation rates, or commodity prices, that could affect the market.
- Data Modeling: Based on this data, the market forecasts are developed for the next 5 years. A combination of trend analysis, scenario modeling, and statistical regression might be used to generate projections.
- Scenario Analysis
- Goal: To test different assumptions and validate how sensitive the market is to changes in key variables (e.g., market demand, regulatory changes, technological disruptions).
- Types of Scenarios:
- Base Case: Based on current assumptions and historical data.
- Best-Case Scenario: Assuming favorable market conditions, regulatory environments, and technological advancements.
- Worst-Case Scenario: Accounting for adverse factors, such as economic downturns, stricter regulations, or unexpected disruptions.