Germany Cardiac Biomarkers Diagnostics Market Report: Trends, Growth and Forecast (2026-2032)
By Product (Reagents & Kits, Instruments/Analyzers, Consumables & Accessories), By Biomarker Type (Troponin (Cardiac Troponin I (cTnI), Cardiac Troponin T (cTnT), High-Sensitivity Troponin (hs-cTn)), Creatine Kinase-MB (CK-MB), Myoglobin, BNP and NT-proBNP, Other Cardiac Biomarkers), By Application (Acute Coronary Syndrome (ACS), Myocardial Infarction (MI), Congestive Heart Failure (CHF), Chest Pain/Emergency Triage, Risk Stratification & Prognosis, Other Cardiac Conditions), By Testing Location (Laboratory Testing, Point-of-Care Testing (POCT)), By End User (Hospitals, Diagnostic Laboratories, Emergency Departments, Cardiac/Specialty Clinics, Ambulatory Care Centers, Others), By Technology (Chemiluminescence Immunoassay, Fluorescence Immunoassay, ELISA, Lateral Flow/Rapid Immunoassay, Other Assay Technologies), By Sample Type (Whole Blood, Plasma, Serum) ... Read more
|
Major Players
|
Germany Cardiac Biomarkers Diagnostics Market Statistics and Insights, 2026
- Market Size Statistics
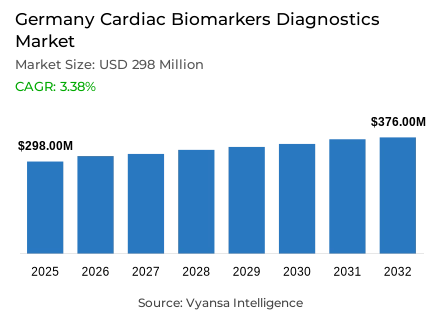
- Cardiac biomarkers diagnostics market size in Germany was valued at USD 298 million in 2025 and is estimated at USD 310 million in 2026.
- The market size is expected to grow to USD 376 million by 2032.
- Market to register a CAGR of around 3.38% during 2026-32.
- Product Shares
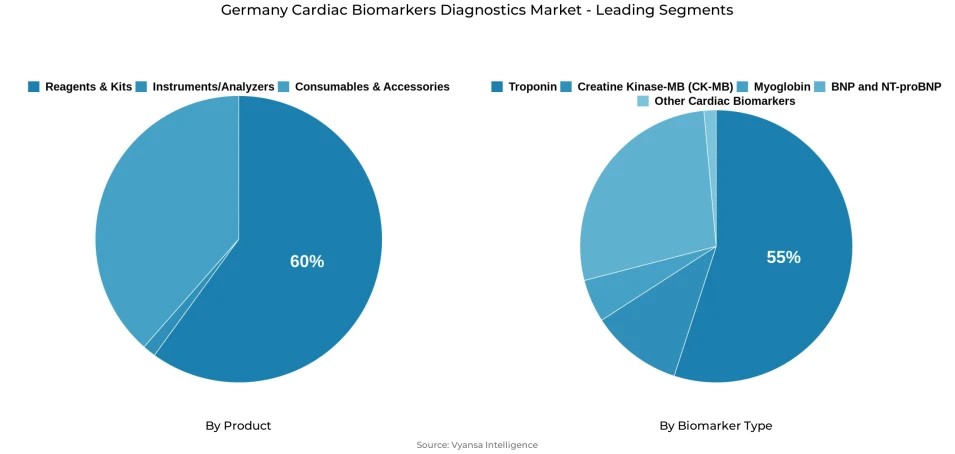
- Reagents & kits grabbed market share of 60%.
- Competition
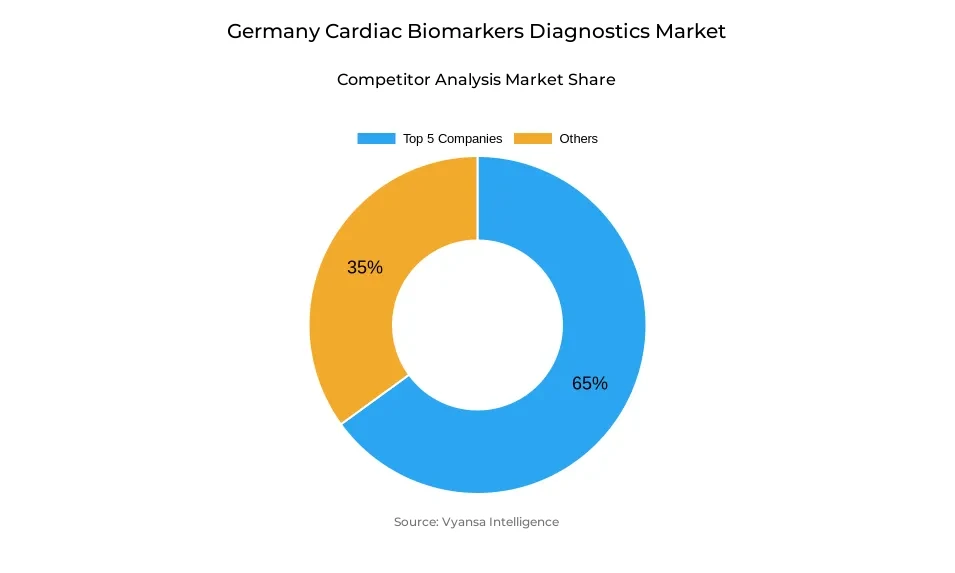
- More than 10 companies are actively engaged in producing cardiac biomarkers diagnostics in Germany.
- Top 5 companies acquired around 65% of the market share.
- bioMérieux, Thermo Fisher Scientific, Ortho Clinical Diagnostics, Abbott Laboratories, Roche Diagnostics etc., are few of the top companies.
- Biomarker Type
- Troponin grabbed 55% of the market.
Germany Cardiac Biomarkers Diagnostics Market Outlook
The Germany cardiac biomarkers diagnostics market size was valued at USD 298 million in 2025 and is projected to grow from USD 310 million in 2026 to USD 376 million by 2032, exhibiting a CAGR of 3.38% during the forecast period. This growth path reflects the steady role of cardiac testing in clinical decision making, where diagnostic accuracy, timely assessment, and routine usage continue to support demand across the healthcare system.
A stable product structure also supports the industry’s direction over the forecast period. Within the overall product landscape, reagents & kits account for around 60% share, showing their strong importance in day to day testing workflows. Since cardiac biomarkers diagnostics relies heavily on repeat consumable usage, this category continues to hold a central place in overall revenue generation and operational demand.
The biomarker mix remains equally important in shaping industry performance. Troponin is almost half of the total biomarker type category at about 55% of the total market share. Its widespread clinical importance in diagnosing the heart, along with its frequent use as a diagnostic tool, makes cardiac biomarkers diagnostic services directly related to existing diagnostic methods, which are already well-known and generally accepted in most case study settings.
As we look to the future, we expect a continued moderate growth trend. Many of the same product types will continue to be in demand, as the primary product categories are directly tied to consistent usage trends. The overall strength of reagents & kits and troponin indicate that cardiac biomarkers diagnostics in Germany will remain based on reliable, regularly used, and clinically significant products, thus ensuring the continued stability of the business outlook through 2032.
Germany Cardiac Biomarkers Diagnostics Market Growth DriverRising Cardiac Burden Keeps Testing Demand Firm
A growing clinical burden continues to support demand for faster and more reliable cardiac assessment across Germany. According to Destatis, chronic ischaemic heart disease caused 70,720 deaths in Germany in 2024, while acute myocardial infarction caused 41,258 deaths in the same year. This level of disease burden keeps hospitals and diagnostic settings focused on timely identification of cardiac events, which directly supports the use of cardiac biomarker testing in routine practice.
The demand base is also reinforced by the country’s ageing profile, which raises the need for regular cardiac evaluation. According to Destatis, 23.2% of Germany’s population was aged 60 to under 80 in 2024, and another 7.2% was aged 80 to under 100. With a large share of the population already in older age groups, the need for cardiac biomarkers diagnostics remains firmly supported by ongoing clinical monitoring and emergency testing requirements.
Germany Cardiac Biomarkers Diagnostics Market ChallengeRegulatory Demands Keep Product Pathways Tight
For diagnostic suppliers in Germany, the ongoing regulatory pressures present a major hurdle in their ability to supply products to customers. Effective 9 July 2024, a New Regulation (EU), Regulation (EUD) 2024/1860, implemented additional obligations to the existing medical device regulations regarding the interruption or discontinuation of the supply of certain IVD(s) set forth under Article 10a for manufacturers of in-vitro diagnostic products. The new regulations require companies to maintain proper documentation of in-vitro diagnostic(s), and, as such, must continually comply with both current and revised IVD(s) regulations, as well as required reporting.
These challenges materialize more clearly as transition timelines begin to move closer. Under the new IVD Regulation, manufacturers of in-vitro diagnostics will need to apply for the necessary approvals to transfer ownership of IVD products with their notified body by either 26 May 2025 (Class D IVDs), 2026 (Class C IVDs), or 2027 (Classes B and A Sterile IVDs) per the EU timeline. This staggered path will create greater demands on documentation, planning, and approvals, resulting in slower response times related to the adjustments of cardiac biomarkers diagnostics in Germany.
Unlock Market Intelligence
Explore the market potential with our data-driven report
Germany Cardiac Biomarkers Diagnostics Market TrendDigital Care Pathways Reshape Test Utilization
A clear market trend is the stronger integration of diagnostics into Germany’s digital care infrastructure. According to the Federal Ministry of Health, the electronic patient record, or ePA, started on 15 January 2025, the nationwide rollout began on 29 April 2025, and all practices, hospitals, and pharmacies have been required to use it since 1 October 2025. This is making it easier to connect test results with broader patient records and treatment pathways.
This shift is already becoming visible in actual system usage. According to gematik, medical facilities recorded peak daily access of about 6 million ePA uses, around 50 million patient records were opened in the first three weeks of nationwide rollout, and about 600,000 documents were uploaded in one week as of 22 May 2025. As digital workflows become more routine, cardiac biomarkers diagnostics is increasingly moving into a more connected and information led care environment.
Germany Cardiac Biomarkers Diagnostics Market OpportunityAgeing Profiles Open Wider Testing Potential
A strong opportunity is developing around the expanding elderly population, which increases the need for repeated cardiovascular assessment. According to Destatis, 23.2% of Germany’s population was aged 60 to under 80 in 2024, while 7.2% was aged 80 to under 100. This means a large share of the population already falls into age groups that more often require regular monitoring, follow up testing, and faster clinical evaluation in both hospital and outpatient settings.
The long term significance of this shift is becoming even clearer. According to Destatis, one in five people in Germany was already aged 67 or older in 2024. This creates room for wider use of cardiac biomarkers diagnostics in older risk populations where early detection and repeat testing become more important in daily care pathways. As patient age profiles continue to reshape service demand, suppliers have a clear opportunity to strengthen testing access, continuity, and clinical relevance across Germany.
Unlock Market Intelligence
Explore the market potential with our data-driven report
Germany Cardiac Biomarkers Diagnostics Market Segmentation Analysis
By Product
- Reagents & Kits
- Instruments/Analyzers
- Consumables & Accessories
Among the product categories, reagents & kits hold the highest share in the Germany cardiac biomarkers biagnostics market, accounting for around 60% of the overall product segment. Their leading position reflects the essential role they play in routine diagnostic workflows, where repeated testing and regular consumable use remain important. Because these products are required in everyday laboratory and clinical testing processes, they continue to form the core of operational demand across the industry.
Their strong share also shows that the product landscape remains closely tied to recurring usage rather than one time system adoption alone. Reagents & Kits support continuous test execution and help maintain the flow of cardiac assessment across different healthcare settings. With such a dominant position already established, this segment continues to act as a major revenue supporting category and remains central to the overall structure and performance of the industry through the forecast period.
By Biomarker Type
- Troponin
- Cardiac Troponin I (cTnI)
- Cardiac Troponin T (cTnT)
- High-Sensitivity Troponin (hs-cTn)
- Creatine Kinase-MB (CK-MB)
- Myoglobin
- BNP and NT-proBNP
- Other Cardiac Biomarkers
Within the biomarker type category, troponin holds the highest share in the Germany cardiac biomarkers diagnostics market, accounting for around 55% of the overall segment. Its leading position shows its continued importance in cardiac related diagnostic use, where reliable biomarker identification remains essential for clinical assessment. This strong share indicates that Troponin stays closely linked with established testing practices and continues to be the most influential biomarker type in the industry.
Its dominance also reflects the preference for diagnostic solutions that are already widely recognized and frequently used in real world testing environments. Since cardiac biomarkers diagnostics depends on clinically trusted markers for regular application, Troponin continues to anchor a significant part of product demand. With more than half of the biomarker type share, the segment remains a key pillar of industry performance and plays an important role in shaping the broader testing landscape in Germany.
List of Companies Covered in Germany Cardiac Biomarkers Diagnostics Market
The companies listed below are highly influential in the Germany cardiac biomarkers diagnostics market, with a significant market share and a strong impact on industry developments.
- bioMérieux
- Thermo Fisher Scientific
- Ortho Clinical Diagnostics
- Abbott Laboratories
- Roche Diagnostics
- Siemens Healthineers
- Danaher Corporation/Beckman Coulter
- Quidel Corporation
- Bio-Rad Laboratories
- Becton, Dickinson and Company (BD)
Market News & Updates
- Roche Diagnostics, 2025:
In September 2025, Roche announced new data for its Elecsys Troponin T hs Gen 6 assay, presented at ESC 2025 and EUSEM 2025, showing that the sixth-generation test can identify acute myocardial infarction and rule out non-AMI cases with high precision; Roche’s Germany diagnostics site also states that the company has been a pioneer in high-sensitivity troponin T and NT-proBNP for more than a decade and is investing in higher-precision diagnostics that simplify rule-in and rule-out pathways and shorten time to discharge. For the Germany cardiac biomarkers market, this is the most material verified update because German emergency departments are highly protocol-driven, so a better-performing next-generation troponin assay from an entrenched supplier can support faster triage, fewer unnecessary downstream tests, and stronger premium positioning for Roche in hospital laboratory and acute-care workflows.
- Siemens Healthineers, 2025:
In April 2025, Siemens Healthineers updated the official documentation for its ADVIA Centaur High-Sensitivity Troponin I assay, confirming its intended use for aiding acute myocardial infarction diagnosis and stating that the assay meets the IFCC definition of a high-sensitivity troponin assay; on its current cardiac-assay pages, Siemens also says its hs-cTnI menu is available across multiple systems including Atellica IM, ADVIA Centaur, Dimension EXL, and Dimension Vista, meets ESC and AACC/IFCC guidance, and can provide 10-minute STAT results on the Atellica Solution. For the Germany cardiac biomarkers market, this matters because Siemens is a domestic medtech leader with a broad installed laboratory base, so continued technical and portfolio reinforcement around hs-cTnI can help laboratories standardize rapid chest-pain pathways, protect incumbent placements, and intensify competition with Roche and Abbott in German hospital diagnostics.
Frequently Asked Questions
Related Report
- Market Segmentation
- Research Scope
- Research Methodology
- Definitions and Assumptions
- Executive Summary
- Germany Cardiac Biomarkers Diagnostics Market Policies, Regulations, and Standards
- Germany Cardiac Biomarkers Diagnostics Market Dynamics
- Growth Factors
- Challenges
- Trends
- Opportunities
- Germany Cardiac Biomarkers Diagnostics Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product
- Reagents & Kits- Market Insights and Forecast 2022-2032, USD Million
- Instruments/Analyzers- Market Insights and Forecast 2022-2032, USD Million
- Consumables & Accessories- Market Insights and Forecast 2022-2032, USD Million
- By Biomarker Type
- Troponin- Market Insights and Forecast 2022-2032, USD Million
- Cardiac Troponin I (cTnI)- Market Insights and Forecast 2022-2032, USD Million
- Cardiac Troponin T (cTnT)- Market Insights and Forecast 2022-2032, USD Million
- High-Sensitivity Troponin (hs-cTn)- Market Insights and Forecast 2022-2032, USD Million
- Creatine Kinase-MB (CK-MB)- Market Insights and Forecast 2022-2032, USD Million
- Myoglobin- Market Insights and Forecast 2022-2032, USD Million
- BNP and NT-proBNP- Market Insights and Forecast 2022-2032, USD Million
- Other Cardiac Biomarkers- Market Insights and Forecast 2022-2032, USD Million
- Troponin- Market Insights and Forecast 2022-2032, USD Million
- By Application
- Acute Coronary Syndrome (ACS)- Market Insights and Forecast 2022-2032, USD Million
- Myocardial Infarction (MI)- Market Insights and Forecast 2022-2032, USD Million
- Congestive Heart Failure (CHF)- Market Insights and Forecast 2022-2032, USD Million
- Chest Pain/Emergency Triage- Market Insights and Forecast 2022-2032, USD Million
- Risk Stratification & Prognosis- Market Insights and Forecast 2022-2032, USD Million
- Other Cardiac Conditions- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location
- Laboratory Testing- Market Insights and Forecast 2022-2032, USD Million
- Point-of-Care Testing (POCT)- Market Insights and Forecast 2022-2032, USD Million
- By End User
- Hospitals- Market Insights and Forecast 2022-2032, USD Million
- Diagnostic Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Emergency Departments- Market Insights and Forecast 2022-2032, USD Million
- Cardiac/Specialty Clinics- Market Insights and Forecast 2022-2032, USD Million
- Ambulatory Care Centers- Market Insights and Forecast 2022-2032, USD Million
- Others- Market Insights and Forecast 2022-2032, USD Million
- By Technology
- Chemiluminescence Immunoassay- Market Insights and Forecast 2022-2032, USD Million
- Fluorescence Immunoassay- Market Insights and Forecast 2022-2032, USD Million
- ELISA- Market Insights and Forecast 2022-2032, USD Million
- Lateral Flow/Rapid Immunoassay- Market Insights and Forecast 2022-2032, USD Million
- Other Assay Technologies- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type
- Whole Blood- Market Insights and Forecast 2022-2032, USD Million
- Plasma- Market Insights and Forecast 2022-2032, USD Million
- Serum- Market Insights and Forecast 2022-2032, USD Million
- By Competitors
- Competition Characteristics
- Market Share & Analysis
- By Product
- Market Size & Growth Outlook
- Germany Reagents & Kits Market Statistics, 2022-2032
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Germany Instruments/Analyzers Market Statistics, 2022-2032
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Germany Consumables & Accessories Market Statistics, 2022-2032
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Biomarker Type- Market Insights and Forecast 2022-2032, USD Million
- By Application- Market Insights and Forecast 2022-2032, USD Million
- By Testing Location- Market Insights and Forecast 2022-2032, USD Million
- By End User- Market Insights and Forecast 2022-2032, USD Million
- By Technology- Market Insights and Forecast 2022-2032, USD Million
- By Sample Type- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Competitive Outlook
- Company Profiles
- Abbott Laboratories
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Roche Diagnostics
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Siemens Healthineers
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Danaher Corporation/Beckman Coulter
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Quidel Corporation
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- bioMérieux
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Thermo Fisher Scientific
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Ortho Clinical Diagnostics
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Bio-Rad Laboratories
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Becton, Dickinson and Company (BD)
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Abbott Laboratories
- Company Profiles
- Disclaimer
| Segment | Sub-Segment |
|---|---|
| By Product |
|
| By Biomarker Type |
|
| By Application |
|
| By Testing Location |
|
| By End User |
|
| By Technology |
|
| By Sample Type |
|
Research Methodology
This study followed a structured approach comprising four key phases to assess the size and scope of the electro-oxidation market. The process began with thorough secondary research to collect data on the target market, related markets, and broader industry context. These findings, along with preliminary assumptions and estimates, were then validated through extensive primary research involving industry experts from across the value chain. To calculate the overall market size, both top-down and bottom-up methodologies were employed. Finally, market segmentation and data triangulation techniques were applied to refine and validate segment-level estimations.
Secondary Research
The secondary research phase involved gathering data from a wide range of credible and published sources. This step helped in identifying industry trends, defining market segmentation, and understanding the market landscape and value chain.
Sources consulted during this phase included:
- Company annual reports, investor presentations, and press releases
- Industry white papers and certified publications
- Trade directories and market-recognized databases
- Articles from authoritative authors and reputable journals
- Gold and silver standard websites
Secondary research was critical in mapping out the industry's value chain and monetary flow, identifying key market segments, understanding regional variations, and tracking significant industry developments.
Other key sources:
- Financial disclosures
- Industry associations and trade bodies
- News outlets and business magazines
- Academic journals and research studies
- Paid industry databases
Primary Research
To validate secondary data and gain deeper market insights, primary research was conducted with key stakeholders across both the supply and demand sides of the market.
On the demand side, participants included decision-makers and influencers from end-user industries—such as CIOs, CTOs, and CSOs—who provided first-hand perspectives on market needs, product usage, and future expectations.
On the supply side, interviews were conducted with manufacturers, industry associations, and institutional participants to gather insights into current offerings, product pipelines, and market challenges.
Primary interviews provided critical inputs such as:
- Market size and revenue data
- Product and service breakdowns
- Market forecasts
- Regional and application-specific trends
Stakeholders consulted included:
- Leading OEM and solution providers
- Channel and distribution partners
- End users across various applications
- Independent consultants and industry specialists
Market Size Estimation and Data Triangulation
- Identifying Key Market Participants (Secondary Research)
- Goal: To identify the major players or companies in the target market. This typically involves using publicly available data sources such as industry reports, market research publications, and financial statements of companies.
- Tools: Reports from firms like Gartner, Forrester, Euromonitor, Statista, IBISWorld, and others. Public financial statements, news articles, and press releases from top market players.
- Extracting Earnings of Key Market Participants
- Goal: To estimate the earnings generated from the product or service being analyzed. This step helps in understanding the revenue potential of each market player in a specific geography.
- Methods: Earnings data can be gathered from:
- Publicly available financial reports (for listed companies).
- Interviews and primary data sources from professionals, such as Directors, VPs, SVPs, etc. This is especially useful for understanding more nuanced, internal data that isn't publicly disclosed.
- Annual reports and investor presentations of key players.
- Data Collation and Development of a Relevant Data Model
- Goal: To collate inputs from both primary and secondary sources into a structured, data-driven model for market estimation. This model will incorporate key market KPIs and any independent variables relevant to the market.
- Key KPIs: These could include:
- Market size, growth rate, and demand drivers.
- Industry-specific metrics like market share, average revenue per customer (ARPC), or average deal size.
- External variables, such as economic growth rates, inflation rates, or commodity prices, that could affect the market.
- Data Modeling: Based on this data, the market forecasts are developed for the next 5 years. A combination of trend analysis, scenario modeling, and statistical regression might be used to generate projections.
- Scenario Analysis
- Goal: To test different assumptions and validate how sensitive the market is to changes in key variables (e.g., market demand, regulatory changes, technological disruptions).
- Types of Scenarios:
- Base Case: Based on current assumptions and historical data.
- Best-Case Scenario: Assuming favorable market conditions, regulatory environments, and technological advancements.
- Worst-Case Scenario: Accounting for adverse factors, such as economic downturns, stricter regulations, or unexpected disruptions.