Brazil Laboratory Information Management System Market Report: Trends, Growth and Forecast (2026-2032)
By Product Type (Clinical Operations LIMS (Hospital Laboratory LIMS, Physician Office Laboratory LIMS, Routine Diagnostics Laboratory LIMS), High-Throughput Testing LIMS (Independent Laboratory LIMS, Commercial Laboratory LIMS, Reference Laboratory LIMS, Multi-site Laboratory Network LIMS), Public Health & Reporting LIMS (Public Health Laboratory LIMS, Surveillance-linked Laboratory LIMS, Reference Network Laboratory LIMS), Research & Repository LIMS (Academic Research Laboratory LIMS, Translational Research Laboratory LIMS, Biobank & Repository LIMS), Regulated QA/QC LIMS (Pharmaceutical Laboratory LIMS, Biotechnology Laboratory LIMS, Food Testing Laboratory LIMS, Environmental Testing Laboratory LIMS, Forensic Laboratory LIMS)), By Workflow Type (Accessioning-to-Result Management (Sample Registration, Sample Tracking, Test Assignment, Result Entry & Release), Instrument-Integrated Workflow Management (Analyzer Connectivity, Automated Data Capture, Bidirectional Instrument Communication), Quality-Controlled Laboratory Workflow (Quality Control Management, Quality Assurance Workflow, Exception & Deviation Handling), Traceability & Repository Workflow (Chain-of-Custody Tracking, Storage & Retrieval Management, Biobank Sample Lifecycle Tracking), Interoperability & Reporting Workflow (External System Connectivity, Standards-based Data Exchange, Regulatory & Public Health Reporting)), By Deployment Model (Controlled Environment Deployment, Scalable Digital Deployment, Mixed Infrastructure Deployment), By End-User Cluster (Care Delivery Laboratories (Hospital Laboratories, Health-System Laboratories, Physician Office Laboratories, Community & Decentralized Testing Laboratories), Commercial Testing Laboratories (Independent Laboratories, Commercial Laboratories, Reference Laboratories), Government & Public Health Laboratories (Public Health Laboratories, Central Reference Laboratories, Disease Surveillance Laboratories), Research & Translational Laboratories (Academic Laboratories, Research Institutes, Biobanks, Clinical Research Support Laboratories), Regulated Scientific & Industrial Laboratories (Pharmaceutical Laboratories, Biotechnology Laboratories, Food Safety Laboratories, Environmental Laboratories, Forensic Laboratories)), By Purchase Model (Perpetual License, Subscription License, Usage-Based Model, Hybrid Commercial Model) ... Read more
|
Major Players
|
Brazil Laboratory Information Management System Market Statistics and Insights, 2026
- Market Size Statistics
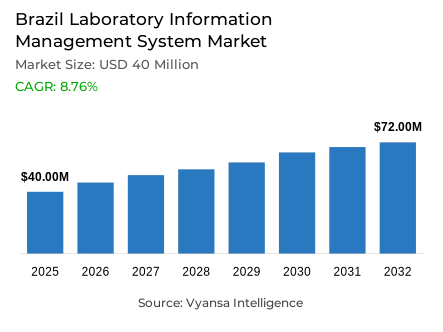
- Laboratory information management system market size in Brazil was valued at USD 40 million in 2025 and is estimated at USD 43 million in 2026.
- The market size is expected to grow to USD 72 million by 2032.
- Market to register a CAGR of around 8.76% during 2026-32.
- Product Type Shares
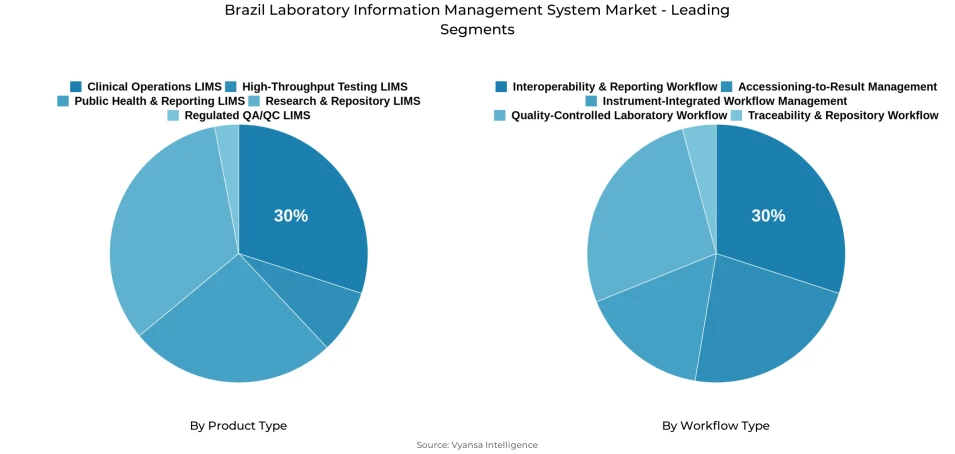
- Clinical operations lims grabbed market share of 30%.
- Competition
- More than 10 companies are actively engaged in producing laboratory information management system in Brazil.
- Top 5 companies acquired around 35% of the market share.
- Abbott, Autoscribe Informatics, Illumina, Thermo Fisher Scientific, Siemens etc., are few of the top companies.
- Workflow Type
- Interoperability & reporting workflow grabbed 30% of the market.
Brazil Laboratory Information Management System Market Outlook
The Brazil Laboratory Information Management System Market was valued at USD 40 million in 2025 and is projected to grow from USD 43 million in 2026 to USD 72 million by 2032, registering a CAGR of 8.76% across the forecast period. This expansion reflects a steady and structured growth phase within the Laboratory Information Management System (LIMS) industry across Brazil, supported by rising demand for laboratory digitization, stronger data handling, and more reliable workflow coordination across clinical and diagnostic environments. Growth is being shaped not by a single catalyst but by a broader institutional shift toward digital accountability and process-driven laboratory management.
At the product level, Clinical Operations LIMS accounts for 30% of the overall market, holding the leading position within the product type segment. This share reflects sustained laboratory demand for systems that manage daily operational activities, routine documentation, and organized sample-related processes with greater precision. Platforms that improve lab data management systems market performance and deliver consistent traceability across testing workflows are being selected with growing regularity as laboratories in Brazil raise their operational standards.
Workflow demand is equally shaped by the Interoperability and Reporting Workflow segment, which commands 30% of the total market. This prominence reflects a rising institutional need to connect data across instruments, systems, and reporting channels without introducing delays or duplication. The digital laboratory solutions market in Brazil continues to benefit from platforms that improve communication quality, streamline data exchange, and support more consistent reporting across laboratory operations at scale.
The overall outlook through 2032 remains favorable as laboratory processes across Brazil become more data-intensive and digitally connected. Clinical execution platforms and interoperability-focused workflows are jointly sustaining adoption momentum, and the laboratory management software market is expected to strengthen its commercial relevance as laboratories invest in smarter, more traceable, and better-connected informatics environments throughout the forecast period.

Brazil Laboratory Information Management System Market Growth Driver
National Health Data Integration Expands LIMS Relevance
Brazil's public health data infrastructure is creating strong and measurable demand for structured laboratory systems across the country. As per data published by the Ministry of Health of Brazil, the Rede Nacional de Dados em Saude became the official interoperability platform of SUS in July 2025, with more than 2.8 billion records already stored, including 75 million exams, 436 million attendances, 30.2 million prescriptions, and 773 million regulation procedures. Managing data flows of this scale requires robust laboratory workflow automation software that can handle larger volumes with stronger traceability and more consistent digital control.
Territorial integration is reinforcing this demand further across Brazil. According to statistics released by the Ministry of Health of Brazil, more than 80% of Brazilian states and 68.3% of municipalities are already integrated into the national platform, with 3,805 municipalities sending records regularly. As more laboratories connect to interoperable public health infrastructure, the need for sample tracking and management systems grows because laboratories must organize test records, reduce manual handling, and support faster data exchange across increasingly complex public health workflows.
Brazil Laboratory Information Management System Market Challenge
Evolving Regulatory Standards Raise Operational Demands
Rising compliance requirements are creating a more demanding operating environment for laboratories and informatics providers across Brazil. Based on data from Anvisa, RDC 978/2025 entered into force on 10 June 2025 and applies to all services performing clinical analysis exams, including laboratories, clinics, pharmacies, and consulting rooms. The agency subsequently published a formal questions-and-answers document in August 2025 to clarify how the new rules should be interpreted and applied, which reflects the operational complexity that these requirements are generating across the sector.
The regulatory burden is intensifying further as the framework continues to evolve. As indicated by authoritative sources at Anvisa, the first edition of the guidance document already incorporated changes introduced by RDC 986, published on 15 August 2025, and RDC 978/2025 also replaces the earlier RDC 786/2023. For laboratories managing data integrity and cybersecurity in labs alongside updated compliance obligations, workflows, audit trails, and reporting routines must be continuously revised in line with newer and more detailed regulatory expectations across Brazil.
Unlock Market Intelligence
Explore the market potential with our data-driven report
Brazil Laboratory Information Management System Market Trend
Molecular Screening Programs Reshape Laboratory Workflows
Organized molecular screening programs are driving a clear and measurable shift in laboratory workflow design across Brazil. According to statistics released by the Ministry of Health of Brazil, the DNA-HPV molecular test was introduced into SUS in August 2025, initially across 12 states, with the test detecting 14 HPV genotypes through a structured screening model rather than isolated opportunistic testing. This organized approach is raising the importance of lab data analytics and reporting tools for managing structured sample movement, result communication, and reporting discipline in routine laboratory operations.
The scope of this trend is expanding as implementation widens nationally. As per data published by the Ministry of Health of Brazil, the first phase is expected to support screening for approximately 5.6 million women across five years in the initial states, while the national target is to benefit 7 million women aged 25 to 64 annually by the end of 2026. As these volumes increase, digital transformation in laboratories across Brazil becomes more commercially urgent because organized molecular screening depends on stronger reporting visibility and workflow coordination at scale.
Brazil Laboratory Information Management System Market Opportunity
Public Laboratory Modernization Unlocks Informatics Investment
Brazil's public laboratory modernization program is generating a significant and tangible commercial opportunity for advanced laboratory informatics solutions. Evidence drawn from public data released by the Ministry of Health of Brazil confirms that R$153 million has already been transferred to the LACEN network for equipment purchases supporting all states, as part of the Novo PAC Saude strategy to modernize laboratories, accelerate diagnoses, and expand access to advanced technologies. This investment is creating direct demand for cloud-based LIMS solutions because improved equipment performance raises the need for better data handling and workflow orchestration across public laboratory networks.
The opportunity is particularly compelling in genomics-intensive environments. Insights sourced from official databases at the Ministry of Health of Brazil indicate that the newly installed MiSeq i100 Plus at LACEN-PR completes sequencing in up to four hours compared to approximately 12 hours previously, supporting analysis of SARS-CoV-2, influenza, meningococcus, tuberculosis, and arboviruses. As public laboratories in Brazil become faster and more data-rich, demand for scalable laboratory management systems capable of managing high-frequency genomic workflows is expected to grow substantially through the forecast period.
Unlock Market Intelligence
Explore the market potential with our data-driven report
Brazil Laboratory Information Management System Market Segmentation Analysis
By Product Type
- Clinical Operations LIMS
- Hospital Laboratory LIMS
- Physician Office Laboratory LIMS
- Routine Diagnostics Laboratory LIMS
- High-Throughput Testing LIMS
- Independent Laboratory LIMS
- Commercial Laboratory LIMS
- Reference Laboratory LIMS
- Multi-site Laboratory Network LIMS
- Public Health & Reporting LIMS
- Public Health Laboratory LIMS
- Surveillance-linked Laboratory LIMS
- Reference Network Laboratory LIMS
- Research & Repository LIMS
- Academic Research Laboratory LIMS
- Translational Research Laboratory LIMS
- Biobank & Repository LIMS
- Regulated QA/QC LIMS
- Pharmaceutical Laboratory LIMS
- Biotechnology Laboratory LIMS
- Food Testing Laboratory LIMS
- Environmental Testing Laboratory LIMS
- Forensic Laboratory LIMS
Clinical Operations LIMS commands the highest share within the product type category at 30%, reflecting sustained institutional demand for platforms that govern routine testing activity, organized record handling, and day-to-day process visibility with operational precision. Laboratories across Brazil are actively prioritizing systems that improve internal coordination and reduce execution gaps, with LIMS for clinical diagnostics laboratories remaining one of the most commercially active product areas within this segment. Its leading position confirms that operational control remains the primary selection criterion in product adoption decisions.
This segment's strength also reflects a market-wide preference for platforms that improve everyday efficiency rather than offering only isolated software functionality. Clinical Operations LIMS is being valued for its ability to support structured workflows, strengthen quality control laboratory software capabilities, and drive more consistent laboratory performance across testing environments in Brazil. As laboratories continue to modernize and expand their operational scope, this segment is expected to maintain its lead position throughout the forecast period.
By Workflow Type
- Accessioning-to-Result Management
- Sample Registration
- Sample Tracking
- Test Assignment
- Result Entry & Release
- Instrument-Integrated Workflow Management
- Analyzer Connectivity
- Automated Data Capture
- Bidirectional Instrument Communication
- Quality-Controlled Laboratory Workflow
- Quality Control Management
- Quality Assurance Workflow
- Exception & Deviation Handling
- Traceability & Repository Workflow
- Chain-of-Custody Tracking
- Storage & Retrieval Management
- Biobank Sample Lifecycle Tracking
- Interoperability & Reporting Workflow
- External System Connectivity
- Standards-based Data Exchange
- Regulatory & Public Health Reporting
The Interoperability and Reporting Workflow segment commands the highest share within the workflow type category at 30%, driven by growing laboratory demand for systems that support smoother data exchange, faster reporting, and better coordination across connected platforms. Laboratories in Brazil are placing increasing value on workflows that reduce process gaps and improve information movement, with regulatory compliance software for labs being integrated as a core component of reporting-first workflow strategies. This segment's lead position confirms that connected reporting capability is a primary driver of workflow adoption decisions.
Its commercial importance is deepening as reporting demands become more complex and interoperability expectations continue to rise. Lab data analytics and reporting tools are being deployed with greater frequency as laboratories across Brazil recognize that reporting quality and cross-system compatibility are now non-negotiable requirements in competitive and compliance-driven environments. As these expectations intensify through the forecast period, this segment is expected to sustain its position as one of the most strategically significant workflow categories within the Brazil Laboratory Information Management System Market.
List of Companies Covered in Brazil Laboratory Information Management System Market
The companies listed below are highly influential in the Brazil laboratory information management system market, with a significant market share and a strong impact on industry developments.
- Abbott
- Autoscribe Informatics
- Illumina
- Thermo Fisher Scientific
- Siemens
- LabVantage Solutions
- LabWare
- Revvity
- LabWorks
- LabLynx
Market News & Updates
- LabVantage Solutions, 2026:
LabVantage launched LabVantage CORTEX, extending its LIMS portfolio with AI, analytics, and automation features. The platform adds agentic AI, cloud-native orchestration, and workflow automation capabilities for laboratory environments, and was introduced by the company during Pittcon 2026 as part of its broader informatics offering.
- Thermo Fisher Scientific, 2026:
Thermo Fisher released updates for SampleManager LIMS v21.3, adding features aimed at workflow efficiency, security, and integration. The version includes token authentication, stronger REST API session handling, and signed proof-of-delivery download capability within the SampleManager laboratory informatics platform.
Frequently Asked Questions
Related Report
- Market Segmentation
- Research Scope
- Research Methodology
- Definitions and Assumptions
- Executive Summary
- Brazil Laboratory Information Management System Market Policies, Regulations, and Standards
- Brazil Laboratory Information Management System Market Dynamics
- Growth Factors
- Challenges
- Trends
- Opportunities
- Brazil Laboratory Information Management System Market Statistics, 2022-2032F
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Product Type
- Clinical Operations LIMS- Market Insights and Forecast 2022-2032, USD Million
- Hospital Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Physician Office Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Routine Diagnostics Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- High-Throughput Testing LIMS- Market Insights and Forecast 2022-2032, USD Million
- Independent Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Commercial Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Reference Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Multi-site Laboratory Network LIMS- Market Insights and Forecast 2022-2032, USD Million
- Public Health & Reporting LIMS- Market Insights and Forecast 2022-2032, USD Million
- Public Health Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Surveillance-linked Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Reference Network Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Research & Repository LIMS- Market Insights and Forecast 2022-2032, USD Million
- Academic Research Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Translational Research Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Biobank & Repository LIMS- Market Insights and Forecast 2022-2032, USD Million
- Regulated QA/QC LIMS- Market Insights and Forecast 2022-2032, USD Million
- Pharmaceutical Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Biotechnology Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Food Testing Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Environmental Testing Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Forensic Laboratory LIMS- Market Insights and Forecast 2022-2032, USD Million
- Clinical Operations LIMS- Market Insights and Forecast 2022-2032, USD Million
- By Workflow Type
- Accessioning-to-Result Management- Market Insights and Forecast 2022-2032, USD Million
- Sample Registration- Market Insights and Forecast 2022-2032, USD Million
- Sample Tracking- Market Insights and Forecast 2022-2032, USD Million
- Test Assignment- Market Insights and Forecast 2022-2032, USD Million
- Result Entry & Release- Market Insights and Forecast 2022-2032, USD Million
- Instrument-Integrated Workflow Management- Market Insights and Forecast 2022-2032, USD Million
- Analyzer Connectivity- Market Insights and Forecast 2022-2032, USD Million
- Automated Data Capture- Market Insights and Forecast 2022-2032, USD Million
- Bidirectional Instrument Communication- Market Insights and Forecast 2022-2032, USD Million
- Quality-Controlled Laboratory Workflow- Market Insights and Forecast 2022-2032, USD Million
- Quality Control Management- Market Insights and Forecast 2022-2032, USD Million
- Quality Assurance Workflow- Market Insights and Forecast 2022-2032, USD Million
- Exception & Deviation Handling- Market Insights and Forecast 2022-2032, USD Million
- Traceability & Repository Workflow- Market Insights and Forecast 2022-2032, USD Million
- Chain-of-Custody Tracking- Market Insights and Forecast 2022-2032, USD Million
- Storage & Retrieval Management- Market Insights and Forecast 2022-2032, USD Million
- Biobank Sample Lifecycle Tracking- Market Insights and Forecast 2022-2032, USD Million
- Interoperability & Reporting Workflow- Market Insights and Forecast 2022-2032, USD Million
- External System Connectivity- Market Insights and Forecast 2022-2032, USD Million
- Standards-based Data Exchange- Market Insights and Forecast 2022-2032, USD Million
- Regulatory & Public Health Reporting- Market Insights and Forecast 2022-2032, USD Million
- Accessioning-to-Result Management- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model
- Controlled Environment Deployment- Market Insights and Forecast 2022-2032, USD Million
- Scalable Digital Deployment- Market Insights and Forecast 2022-2032, USD Million
- Mixed Infrastructure Deployment- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster
- Care Delivery Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Hospital Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Health-System Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Physician Office Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Community & Decentralized Testing Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Commercial Testing Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Independent Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Commercial Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Reference Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Government & Public Health Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Public Health Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Central Reference Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Disease Surveillance Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Research & Translational Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Academic Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Research Institutes- Market Insights and Forecast 2022-2032, USD Million
- Biobanks- Market Insights and Forecast 2022-2032, USD Million
- Clinical Research Support Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Regulated Scientific & Industrial Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Pharmaceutical Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Biotechnology Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Food Safety Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Environmental Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Forensic Laboratories- Market Insights and Forecast 2022-2032, USD Million
- Care Delivery Laboratories- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model
- Perpetual License- Market Insights and Forecast 2022-2032, USD Million
- Subscription License- Market Insights and Forecast 2022-2032, USD Million
- Usage-Based Model- Market Insights and Forecast 2022-2032, USD Million
- Hybrid Commercial Model- Market Insights and Forecast 2022-2032, USD Million
- By Competitors
- Competition Characteristics
- Market Share & Analysis
- By Product Type
- Market Size & Growth Outlook
- Brazil Clinical Operations LIMS Market Statistics, 2022-2032
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Workflow Type- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Brazil High-Throughput Testing LIMS Market Statistics, 2022-2032
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Workflow Type- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Brazil Public Health & Reporting LIMS Market Statistics, 2022-2032
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Workflow Type- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Brazil Research & Repository LIMS Market Statistics, 2022-2032
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Workflow Type- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Brazil Regulated QA/QC LIMS Market Statistics, 2022-2032
- Market Size & Growth Outlook
- By Revenues in USD Million
- Market Segmentation & Growth Outlook
- By Workflow Type- Market Insights and Forecast 2022-2032, USD Million
- By Deployment Model- Market Insights and Forecast 2022-2032, USD Million
- By End-User Cluster- Market Insights and Forecast 2022-2032, USD Million
- By Purchase Model- Market Insights and Forecast 2022-2032, USD Million
- Market Size & Growth Outlook
- Competitive Outlook
- Company Profiles
- Thermo Fisher Scientific
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Siemens
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- LabVantage Solutions
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- LabWare
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Revvity
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Abbott
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Autoscribe Informatics
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Illumina
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- LabWorks
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- LabLynx
- Business Description
- Product Portfolio
- Collaborations & Alliances
- Recent Developments
- Financial Details
- Others
- Thermo Fisher Scientific
- Company Profiles
- Disclaimer
| Segment | Sub-Segment |
|---|---|
| By Product Type |
|
| By Workflow Type |
|
| By Deployment Model |
|
| By End-User Cluster |
|
| By Purchase Model |
|
Research Methodology
This study followed a structured approach comprising four key phases to assess the size and scope of the electro-oxidation market. The process began with thorough secondary research to collect data on the target market, related markets, and broader industry context. These findings, along with preliminary assumptions and estimates, were then validated through extensive primary research involving industry experts from across the value chain. To calculate the overall market size, both top-down and bottom-up methodologies were employed. Finally, market segmentation and data triangulation techniques were applied to refine and validate segment-level estimations.
Secondary Research
The secondary research phase involved gathering data from a wide range of credible and published sources. This step helped in identifying industry trends, defining market segmentation, and understanding the market landscape and value chain.
Sources consulted during this phase included:
- Company annual reports, investor presentations, and press releases
- Industry white papers and certified publications
- Trade directories and market-recognized databases
- Articles from authoritative authors and reputable journals
- Gold and silver standard websites
Secondary research was critical in mapping out the industry's value chain and monetary flow, identifying key market segments, understanding regional variations, and tracking significant industry developments.
Other key sources:
- Financial disclosures
- Industry associations and trade bodies
- News outlets and business magazines
- Academic journals and research studies
- Paid industry databases
Primary Research
To validate secondary data and gain deeper market insights, primary research was conducted with key stakeholders across both the supply and demand sides of the market.
On the demand side, participants included decision-makers and influencers from end-user industries—such as CIOs, CTOs, and CSOs—who provided first-hand perspectives on market needs, product usage, and future expectations.
On the supply side, interviews were conducted with manufacturers, industry associations, and institutional participants to gather insights into current offerings, product pipelines, and market challenges.
Primary interviews provided critical inputs such as:
- Market size and revenue data
- Product and service breakdowns
- Market forecasts
- Regional and application-specific trends
Stakeholders consulted included:
- Leading OEM and solution providers
- Channel and distribution partners
- End users across various applications
- Independent consultants and industry specialists
Market Size Estimation and Data Triangulation
- Identifying Key Market Participants (Secondary Research)
- Goal: To identify the major players or companies in the target market. This typically involves using publicly available data sources such as industry reports, market research publications, and financial statements of companies.
- Tools: Reports from firms like Gartner, Forrester, Euromonitor, Statista, IBISWorld, and others. Public financial statements, news articles, and press releases from top market players.
- Extracting Earnings of Key Market Participants
- Goal: To estimate the earnings generated from the product or service being analyzed. This step helps in understanding the revenue potential of each market player in a specific geography.
- Methods: Earnings data can be gathered from:
- Publicly available financial reports (for listed companies).
- Interviews and primary data sources from professionals, such as Directors, VPs, SVPs, etc. This is especially useful for understanding more nuanced, internal data that isn't publicly disclosed.
- Annual reports and investor presentations of key players.
- Data Collation and Development of a Relevant Data Model
- Goal: To collate inputs from both primary and secondary sources into a structured, data-driven model for market estimation. This model will incorporate key market KPIs and any independent variables relevant to the market.
- Key KPIs: These could include:
- Market size, growth rate, and demand drivers.
- Industry-specific metrics like market share, average revenue per customer (ARPC), or average deal size.
- External variables, such as economic growth rates, inflation rates, or commodity prices, that could affect the market.
- Data Modeling: Based on this data, the market forecasts are developed for the next 5 years. A combination of trend analysis, scenario modeling, and statistical regression might be used to generate projections.
- Scenario Analysis
- Goal: To test different assumptions and validate how sensitive the market is to changes in key variables (e.g., market demand, regulatory changes, technological disruptions).
- Types of Scenarios:
- Base Case: Based on current assumptions and historical data.
- Best-Case Scenario: Assuming favorable market conditions, regulatory environments, and technological advancements.
- Worst-Case Scenario: Accounting for adverse factors, such as economic downturns, stricter regulations, or unexpected disruptions.